Introduction
Wrist ligament injuries represent a common diagnostic and therapeutic challenge in clinical orthopedics and sports medicine practice. Among the carpal ligaments, the scapholunate ligament (SLL) and the luno-triquetral ligament (LTL) are the most frequently injured.1 However, LTL injuries are often more complex in presentation and more easily overlooked.2,3 A tear of the LTL leads to widening of the lunotriquetral joint space and alters the kinematics of the proximal carpal row. Over time, this may result in volar intercalated segment instability (VISI) and eventually pan-carpal arthritis, producing chronic wrist pain and severe functional limitation. This degenerative cascade progresses more rapidly and severely when associated with concomitant injuries of extrinsic stabilizing ligaments.4,5
Current management strategies include conservative measures or surgical interventions such as temporary pin fixation, direct repair, ligament reconstruction, or arthrodesis depending on individual demands and LTL injured circumstances.3,6,7 With the chronic injury and high functional demand patients, anatomic reconstruction of the ligament is the primary surgical goal to restore maximal wrist stability and motion.8–10 However, the aim of the previously discussed reconstructive procedures using the extensor carpi ulnaris (ECU) or flexor carpi ulnaris (FCU) restored the carpal kinematics of the lunotriquetral joint—rather than its anatomy.11,12 The ligament itself is not rebuilt in this way, and even while the carpal kinematics are better, they cannot precisely mimic those of the native ligament.13 As a result, the rate of reconstructive surgery fail was high up to 31,4% of cases.9 This may be due to failure to restore the original anatomical structure of the ligament and its biomechanical properties. Therefore, detailed understanding of the anatomical structure and function of each component of the LTL is necessary to improve future reconstructive surgery outcomes.
Over the past two decades, several anatomical studies have advanced the understanding of the LTL, contributing to improvements in diagnosis and surgical interventions. However, most studies have described only the general morphology of the ligament, with limited detailed quantitative analysis of each component.14–18 This knowledge gap limits the precision of anatomical ligament reconstruction techniques. In this study, we aimed to reinforce the entire knowledge of the anatomical morphology, add-in the detailed dimensions of LTL and suggest a new surgical technique for LTL reconstruction.
Materials and Methods
Ethics approval
The Institutional Review Board of the University of Medicine and Pharmacy at Ho Chi Minh City approved this research, number: 409/HĐĐĐ-ĐHYD. All human-related procedures complied with institutional and/or national ethics committee standards and followed the 1964 Helsinki Declaration and its later updates or equivalent ethical norms.
This study employed a descriptive case-series design. Dissections were performed on 32 fresh frozen cadaveric wrists from 16 specimens at the Department of Anatomy, University of Medicine and Pharmacy at Ho Chi Minh City. Additionally, one specimen with two wrists was used to demonstrate a new surgical technique for LTL reconstruction.
Exclusion criteria
Specimens with gross evidence of prior trauma, surgery, infection, tumor, severe degenerative changes, or congenital anomalies that might alter wrist anatomy.
Dissection Process
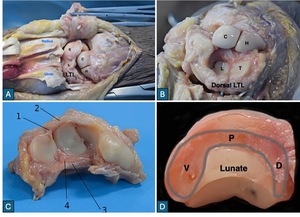
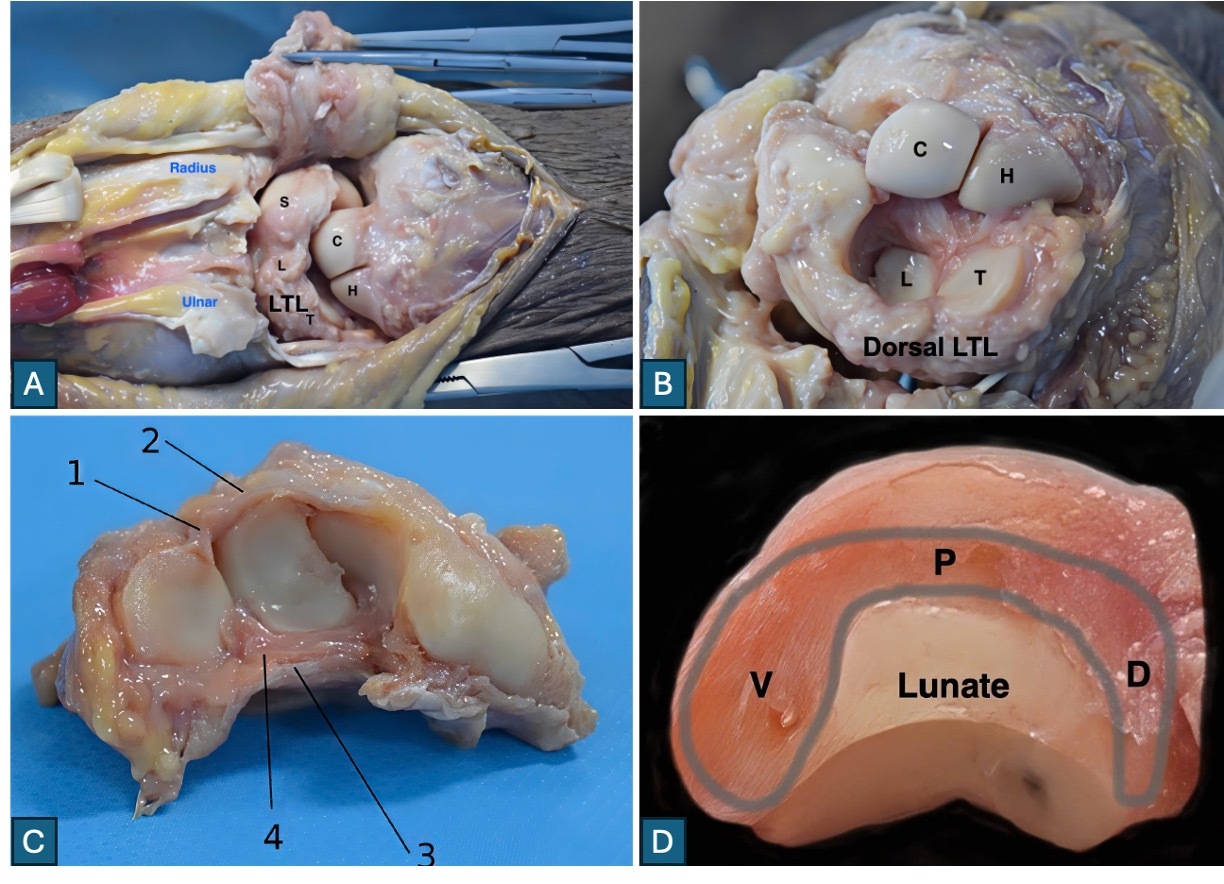
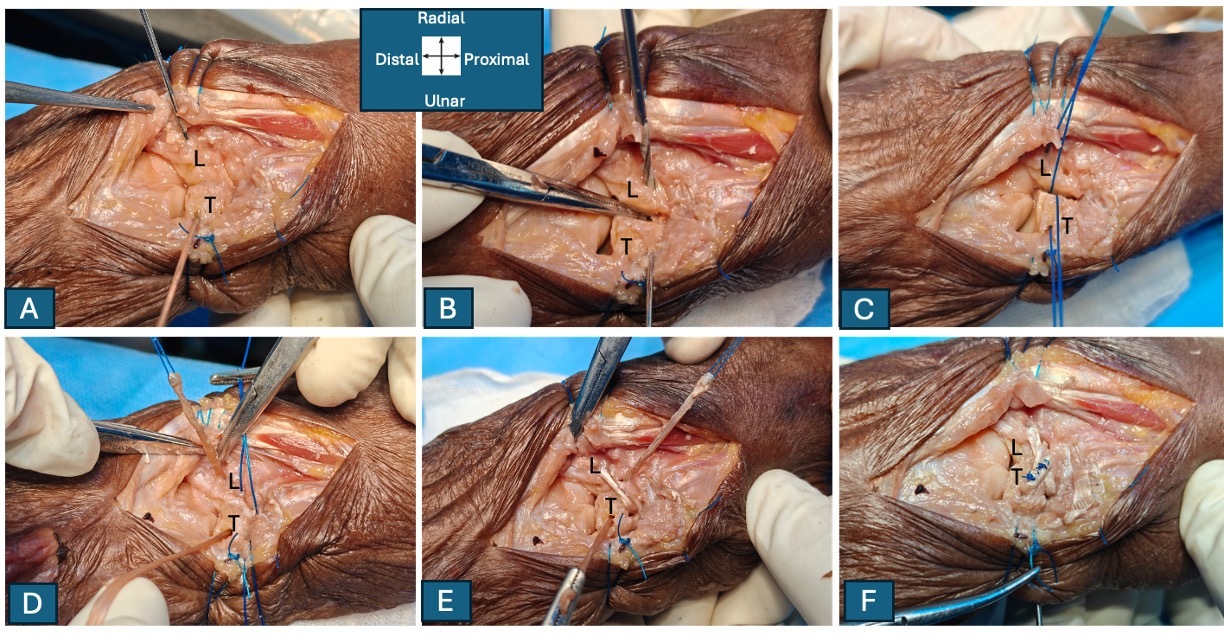
All dissections and measurements were performed by a single orthopedic surgeon with 20 years of experience. The cadavers were thawed and dissected within 3–4 hours to preserve ligament quality. At first, wrists were placed in pronation. A dorsal midline incision (~10 cm) was made from the middle of the third metacarpal proximally to the distal forearm. Following reflection of skin and subcutaneous tissue, the extensor retinaculum was incised through the fourth extensor compartment, and the extensor digitorum and indicis were retracted medially. The dorsal capsule and dorsal radiocarpal and intercarpal ligaments were exposed. The posterior capsule was incised in a U-shape to visualize the proximal carpal row and dorsal interosseous ligaments (Figure 1A). Then, the distal carpal row was separated and palmarly flexed to expose the whole of lunotriquetral joint (Figure 1B). Next, the wrists were turned into full supination. A volar approach was also performed, including division of the transverse carpal ligament and mobilization of flexor tendons and the median nerve to expose the volar capsule. Careful capsulotomy along the radiocarpal and midcarpal joints allowed en bloc removal of the scaphoid–lunate–triquetrum complex. The pisiform was excised, leaving the intact lunotriquetral joint and LTL complex for detailed observation and measurement (Figure 1C).
Gross morphology was assessed macroscopically, with a 4X surgical loupe, and dimensions were recorded with a digital caliper (Insize; accuracy 0.03 mm). Recorded anatomical features included: presence of volar, proximal, and dorsal components; continuity between components; ligament’s origin and insertion, joint-surface configuration; and capsular relationships.
Measurements
-
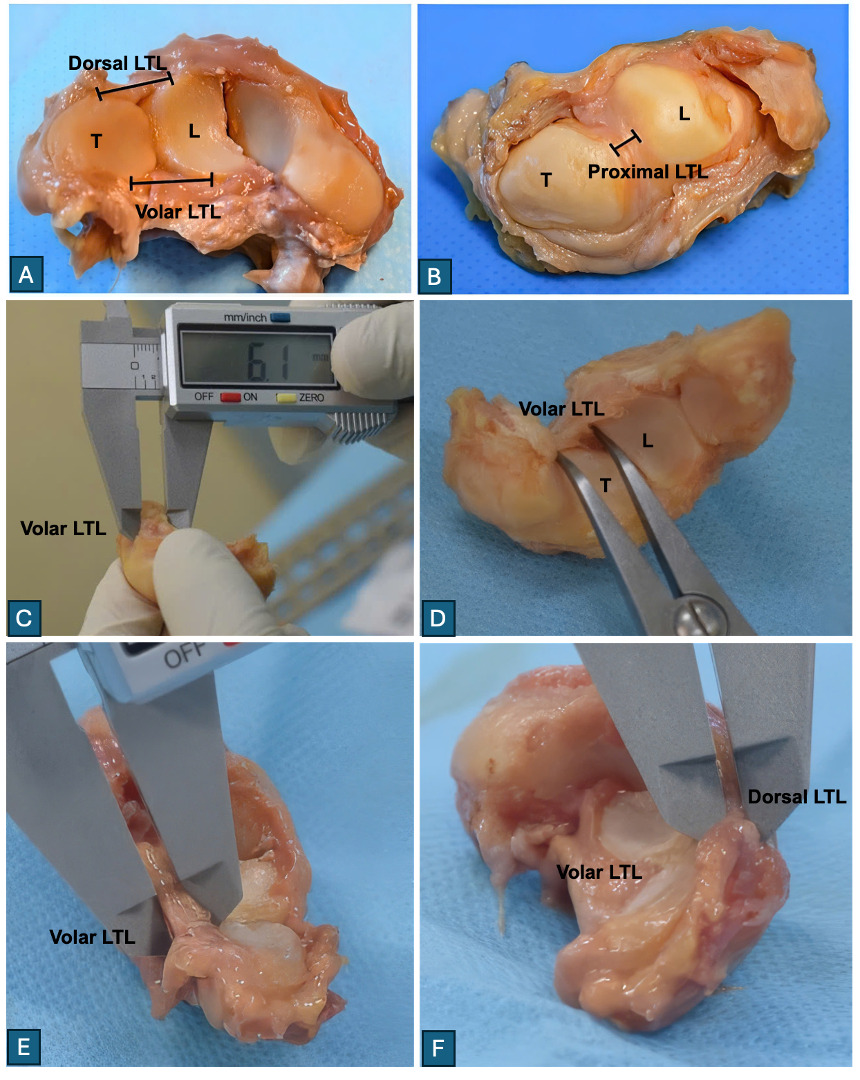
Length: distance between bony insertions of each component (volar, proximal, dorsal) (Figure 2A, 2B, 2D).
-
Width: measured perpendicularly across each component at defined points (Figure 2C).
-
Thickness: assessed on sagittal sections through the lunotriquetral joint (Figure 2E, 2F).
The length of the Volar portion was defined as the distance between the attachment points on the lunate and triquetrum at the anteroinferior corner of the joint. The length of the Proximal portion was measured as the distance between the centers of the attachment surfaces on the dorsal aspect of both bones. The length of the Dorsal portion was defined as the distance between the attachment points on both bones at the posteroinferior corner of the joint.
The width of the Volar portion was measured from the superior to the inferior edge of the ligament on the volar surface. The width of the Proximal portion was defined as the distance from the anterior margin (transition to the volar portion) to the posterior margin (transition to the dorsal portion) on the articular surface. The width of the Dorsal portion was measured from the superior to the inferior edge of the ligament on the dorsal surface.
The thickness was assessed on cross-sections after splitting the lunotriquetral joint along the sagittal plane. The thickness of each portion (volar, proximal, dorsal) was defined as the breadth of the ligament in its respective section.
Data were entered into Microsoft Excel 2013 and analyzed using R version 3.5.1. Qualitative variables were expressed as frequency and percentage. Quantitative variables were presented as mean ± SD. Independent sample t-tests (or Wilcoxon test for non-normal distributions) were used to compare ligament dimensions between components and between sexes. A p-value < 0.05 was considered statistically significant.
Results
The study analyzed 32 cadaveric wrists (18 males [56%], 14 females [44%]). The mean specimen age was 73.9 years old, with 88% obtained from individuals older than 65 years old. No specimen demonstrated prior trauma, surgery, or pathological alterations of wrist anatomy.
Gross morphology
The LTL was consistently identified as a distinct, C-shaped ligament composed of three continuous components—volar, proximal, and dorsal—in 100% of specimens (Figure 1D). Each component exhibited distinct morphological characteristics. No cases of congenital fusion or anomalous variants were observed.
-
Volar component: A thick, robust structure covered by volar capsule in all specimens. Fibers thickened centrally and tapered toward articular margins. In 28 of 32 specimens, triangular thickening was observed at the anteroinferior joint margin. The distal fibers consistently interdigitated with the volar component of the SLL. Its origin was from the anteromedial margin of the lunate, inserting onto the anterolateral margin of the triquetrum. On sagittal sectioning, it was the thickest component of the LTL.
-
Proximal component: A thin connective band located dorsally on the lunotriquetral articular surface, not covered by joint capsule in any specimen. It originated from the superior-medial aspect of the lunate and inserted on the superior-lateral aspect of the triquetrum. Though macroscopically the thinnest component, 27/32 specimens showed central thickening on sagittal section.
-
Dorsal component: A long band partially covered by the thin dorsal capsule. Its fibers were distinct and separate from the dorsal radiocarpal ligament and dorsal SLL. Central thickening was observed, with triangular thickening at the posteroinferior joint margin in 27/32 specimens. It originated from the posteromedial lunate and inserted onto the posterolateral triquetrum. While thicker than the proximal component, it remained thinner than the volar component.
Quantitative analysis
The length of the three parts of the LTL is less than the width (p<0.001). The volar width, dorsal length, width and thickness, and proximal length and thickness of the male wrist group were statistically larger than those of the female wrist group (p<0.05).
-
Length & thickness: The volar component was significantly longer and thicker than both the dorsal and proximal components (p < 0.01). The dorsal component was also significantly longer and thicker than the proximal component (p < 0.01).
-
Width: The proximal component was significantly the widest (p < 0.01). No significant difference in width was found between the volar and dorsal components (p > 0.05).
These findings confirm the morphometric predominance of the volar component, highlighting its likely biomechanical importance in maintaining lunotriquetral joint stability.
New surgical technique for LTL reconstruction
To accommodate the morphometric dimensions of the LTL complex, the palmaris longus tendon (PLT) was selected as a suitable autograft. We developed a dorsal-approach technique for LTL reconstruction utilizing double V shaped bone tunnels (Figure 3). Two separate 2.0 mm bone tunnels were created in each of the lunate and triquetrum, maintaining a safe bone bridge to minimize the risk of fracture. The first V tunnel is positioned in the proximal portion of the bone, while the second V tunnel is located distally. On the triquetrum, the drilling trajectory proceeds obliquely from dorsoulnar to volar radial, exiting at the anterior border of the lunotriquetral joint. On the lunate, the drilling direction runs from dorsoradial to ulnar–volar, exiting at the anterior margin of the lunotriquetral joint, symmetrically oriented to the tunnel created in the triquetrum. A 1.0 nylon suture was then passed through each tunnel to shuttle the PLT graft, allowing it to traverse continuously through the tunnels and form double volar tendon bands spanning the lunotriquetral joint. The graft ends were crossed dorsally and secured within the bone tunnels using appropriate anchors. Finally, the two dorsal graft limbs were sutured together to reinforce the reconstruction.
Discussion
Our study provides the first detailed anatomical dataset of the luno-triquetral ligament (LTL) complex in the Vietnamese population. Our results consistently confirm the three-part architecture of the ligament and establish key morphometric parameters, highlighting the structural predominance of the volar portion. The findings highlight both the complexity of the LTL’s subregions and the challenges in restoring native anatomy, kinematics and extending prior work in this area.
Macroscopically, in all specimens the LTL comprises three continuous but distinct components—volar, proximal, and dorsal—forming a characteristic C-shaped structure around the lunotriquetral joint. These observations are consistent with the previous descriptions, confirming the reproducibility of this three-part configuration across populations.6,17,19 The anatomical relationships are also reaffirmed: the volar portion is reinforced by the volar capsule, the proximal portion is always uncovered by capsule, and the dorsal portion runs independently yet closely related to other dorsal wrist ligaments.
Morphometric analysis provides the most clinically relevant insights. Our study demonstrates that the volar portion is the thickest and longest ligament, whereas the proximal portion is the widest one. The dominance of the volar component in terms of thickness and length has also been reported by Nagao, Lee, and Ritt.5,14,18 This is of particular interest when compares with the scapholunate ligament (SLL), another key interosseous ligament of the proximal carpal row. While the dorsal SLL has long been recognized as biomechanically superior, the LTL exhibits opposite characteristics with the volar component as its structurally dominant portion, acts as the primary restriction on lunotriquetral mobility.11,15 Kinematic study has shown that both the lunate and triquetrum are flexed by sectioning of the palmar LTL leading to VISI deformity.5 This contrast highlights fundamental differences between the radial and ulnar columns of the wrist, emphasizing the central role of the volar LTL in stabilizing the lunotriquetral joint.20,21 The volar LTL contributes primarily to axial load resistance and palmar stability, particularly during wrist flexion–extension and ulnar deviation.6 In contrast, the dorsal LTL provides essential rotational control between the lunate and triquetrum and is considered the key restraint against volar intercalated segment instability (VISI).22
Clinically, these findings emphasize that applying the same reconstructive principles to both SLL and LTL is anatomically inappropriate. Techniques that focus solely on the dorsal portion, as reported in some reconstructions, may fail to restore the natural biomechanics of the lunotriquetral joint.12,13 Shin et al. described an LTL reconstruction technique using a distally based strip of the extensor carpi ulnaris (ECU) tendon, which was inserted into bone tunnels created in the triquetrum and lunate, and then sutured back to its native insertion.9 However, the study did not clearly specify the bone tunnel dimensions or the size of the tendon graft. Subsequently, Giladi et al. detailed a refined surgical method that employed a 3-mm ECU tendon graft and 2.7-mm bone tunnels.11 This single tendon graft was unable to re-establish the full width of both the volar and dorsal components of the LTL ligament. Additionally, in this technique, the tunnels in each bone were drilled from the lunotriquetral joint outward, in a proximal-to-distal direction. Because the drilling trajectory could not extend anteriorly toward the palmar margins of the lunate and triquetrum, reconstruction of the volar component is not feasible. As a result, these reconstruction methods may restore carpal kinematics rather than replicate true LTL anatomy.13 Furthermore, when the ECU tendon is mobilized at the level of the ulnar head, the tendon itself may be directly injured, the subsheath may be disrupted, and the tendon may become unstable within the characteristically shallow ECU groove.23
Harper published firstly the LTL reconstruction technique using a PLT autograft, employing a 3 mm V pattern tunnel drilled from the central dorsal aspect of each bone toward the anterosuperior corner.13 This method encountered similar limitations, as the graft diameter could not reproduce the native width of the lunotriquetral ligament. Moreover, a single tendon strip is unlikely to adequately restore the rotational stability required for normal lunotriquetral joint mechanics. Based on our data, we recommend technical modifications ensuring accurate graft placement and adequate graft size, with need a close attention to restoring the volar portion. For that, we propose an optimal graft thickness and width at least 2 mm and 6.5 mm, respectively, for the volar component and 1.2 mm and 6.3 mm for the dorsal component. To meet these requirements, the PLT would be an ideal graft for LTL reconstruction, with an average diameter of approximately 3 mm and an average length of approximately 14 cm.24 The diameter of the PLT is compatible with the thickness of the volar portion of the LTL. However, both the dorsal and volar components of the LTL have a width of approximately more than 6 mm, corresponding to double bands of the PLT. Therefore, instead of creating a single large bone tunnel, we propose drilling two smaller tunnels at the proximal and distal portions of each bone while preserving an adequate bone bridge to minimize the risk of fracture. Reconstruction of the LTL using double tendon bands may enhance overall stability and provide superior rotational control compared with a single-strand graft.
Despite the strengths of our anatomical dataset, several limitations merit attention. Our study includes the relatively advanced mean age of the cadaveric specimens, which may not fully represent the younger population typically affected by traumatic LTL injuries. Moreover, this was an anatomy-based investigation, and ligament quality in cadaveric tissue cannot be directly compared with that of living tissue. This may affect the measurement results. While our findings highlight the anatomical predominance of the volar component of the LTL, dynamic kinematic studies are still needed to clarify the functional contribution of each ligamentous portion. The four-tunnel (2.0-mm) LTL reconstruction technique demonstrated initial feasibility in cadaveric testing; however, its true effectiveness requires clinical validation. In addition, biomechanical comparisons between single-band and double-band tendon graft reconstructions remain necessary to determine the optimal surgical approach.
Conclusion
In sum, our anatomical study defines the complex structure of the LTL and offers concrete data to inform surgical reconstruction, in which the volar portion of the LTL as the morphometrically superior component between distinct three parts. Surgical reconstruction techniques should therefore prioritize restoration of the volar portion with regard to its width and thickness to re-establish carpal stability. Besides, we also propose the modified LTL reconstruction method using double bands of PLT autograft. However, continued work, including biomechanical modeling and clinical outcome studies, is needed to translate these anatomical insights into improved patient outcomes.
Acknowledgments
We sincerely thank the Department of Anatomy, School of Medicine, University of Medicine and Pharmacy at Ho Chi Minh City for their assistance in specimen preparation, imaging, and access to laboratory facilities, all of which were essential to completing this study.
Authors’ contributions
Quyen Ngoc Le: Conceptualization, Methodology, Software, Resources, Writing - Review & Editing. Hieu Trung Nguyen: Conceptualization, Formal analysis, Data curation, Writing - Review & Editing, Funding acquisition. Minh Dung Nguyen: Conceptualization, Methodology, Investigation, Writing- Original draft preparation. Hung Phuoc Do: Supervision, Review & Editing.
Conflicting interests
The authors declare that they have no conflicts of interest related to this article.
Funding
No specific grant from any funding agency in the public, commercial, or not-for-profit sectors was received for this study.
Informed consent
Informed consent was not applied for the present research because it was a cadaveric study.
Ethical approval
Ethical approval was obtained at University of Medicine and Pharmacy at Ho Chi Minh City, number: 409/HĐĐĐ-ĐHYD. This study was conducted in accordance with the ethical standards of the Declaration of Helsinki (1964) and its subsequent amendments.