Introduction
Osteoporotic vertebral compression fractures (OVCFs) are the most common fragility fractures in older adults, which affects over 1.5 million patients in the United States alone each year.1 With the global population aging, these numbers will continue to grow. OVCFs are often associated with sudden back pain and progressive kyphotic deformity, increasing the risk of future vertebral and hip fractures.2 Conservative treatment such as analgesia or bracing can relieve symptoms in some patients but usually does not provide adequate pain control or early mobilization.3,4
Over the past two decades, percutaneous vertebroplasty (PVP) and percutaneous kyphoplasty (PKP) have become widely adopted minimally invasive options for patients with persistent pain or instability after an acute osteoporotic fracture.3,5These techniques have achieved rapid pain reduction in most of the patients and often within days of the procedure. In addition, these procedures help restore spinal alignment by stabilizing the collapsed vertebral body and partially recovering lost height.6 As a result, vertebral augmentation is now considered a key component of modern management for symptomatic OVCFs, offering faster recovery and improved quality of life compared with prolonged conservative care.
Despite the well-documented effectiveness of vertebral augmentation for acute osteoporotic fractures, a subset of patients experience recurrent pain due to new vertebral fractures occurring at the treated level or adjacent levels.7,8 Previous studies have reported vertebral refracture rates of up to approximately 35% of treated cases.7,8 These events are thought to result from progressive osteoporosis, altered spinal biomechanics, and increased stress at neighboring endplates after cement injection.9,10 Although repeat vertebral augmentation is often performed in these cases, the available evidence is mostly limited to small retrospective series or case reports with variable methodology and follow-up. As a result, the true clinical benefit, radiologic correction, and complication profile of repeat vertebral augmentation remain unclear.
Therefore, this systematic review and meta-analysis aimed to synthesize the available evidence on repeat vertebral augmentation for recurrent or adjacent OVCFs. The primary objective was to evaluate clinical outcomes in pain and function, radiologic parameters, and procedural aspects to provide a comprehensive overview of the efficacy and safety of re-augmentation in this patient population.
Methods
Study Design
This systematic review and meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. The protocol was prospectively registered in the PROSPERO database (ID: CRD420251171534). The aim of this study was to summarize and evaluate the available evidence on the clinical, radiologic, and procedural outcomes of repeat (revision) PVP or PKP performed for recurrent or adjacent OVCFs.
Literature Search
A comprehensive literature search was performed in PubMed, Scopus, and Web of Science up to October 28th, 2025. The search combined keywords related to vertebral augmentation (vertebroplasty, kyphoplasty, vertebral augmentation) and repeat procedures (redo, revision, re-augmentation, re-injection). Reference lists of relevant reviews and included studies were also screened to identify any additional eligible articles. Only English-language human studies were considered. Conference abstracts, technical notes, and biomechanical or cadaveric investigations were excluded, as were studies addressing traumatic, metastatic, or infectious vertebral lesions.
Eligibility Criteria
Studies were included if they reported repeat PVP or PKP for recurrent or adjacent OVCFs, enrolled at least five patients, and provided pre- and post-procedure data on pain, function, or radiologic outcomes. Studies describing only primary (non-repeat) augmentation, non-osteoporotic or metastatic fractures, or those lacking sufficient outcome data were excluded. In addition, studies were excluded if they did not report sufficient continuous data of pre- and post-operative outcomes (e.g., VAS, ODI), or where the endpoint was a binary complication rather than mean change.
Study Selection and Data Extraction
Two reviewers independently screened all titles, abstracts, and full texts. Any disagreements were resolved through discussion and consensus. Studies were grouped according to the type of repeat procedure (PVP, PKP, or mixed) for pooled analyses. Data extraction was performed using a standardized spreadsheet and included study details and all reported outcome measures. Clinical data included pain scores measured by the Visual Analogue Scale (VAS) or Numerical Rating Scale (NRS) and functional outcomes assessed by the Oswestry Disability Index (ODI). Radiologic data comprised segmental kyphotic angle (SKA) and vertebral height loss (VHL). When standard deviations were unavailable, they were estimated from reported confidence intervals or ranges using established methods. Pain scores reported on the NRS were converted to equivalent 10-point VAS values for consistency across studies. Procedural characteristics such as cement volume during the initial and repeat augmentation and the interval between procedures were also recorded. In addition, all reported complications, such as cement leakage, adjacent or same-level refracture, infection, neurological deficit, and need for re-revision, were extracted for analysis.
Risk of Bias Assessment
The methodological quality of all included studies was assessed using the Risk of Bias In Non-randomized Studies of Interventions (ROBINS-I) tool.11 Each domain was rated as low, moderate, or serious risk of bias by two independent reviewers, with discrepancies resolved by consensus. Potential reporting bias was considered and publication bias was separately evaluated using funnel plot symmetry and Egger’s regression test.
Statistical Analysis
All statistical analyses were conducted using R software (version 4.5.1). Continuous outcomes (VAS, ODI, SKA, and cement volume) were synthesized as mean differences (MDs) using a random-effects model according to the DerSimonian–Laird method. Proportion outcomes (cement leakage, adjacent-level fracture, and re-revision) were pooled using the Freeman–Tukey double-arcsine transformation under a random-effects model, with inverse-variance weighting applied to all proportion analyses. Statistical heterogeneity was quantified using the I² and τ² statistics, with I² values greater than 50 % considered indicative of substantial heterogeneity. Publication bias was explored visually with funnel plots and quantitatively with Egger’s regression test when at least ten studies were available. A two-sided p-value of less than 0.05 was considered statistically significant.
Data Presentation
Baseline characteristics of the included studies are summarized in Table 1.12–25 Pooled quantitative outcomes for clinical, radiologic, and complication endpoints are presented in Tables 2 and 3 and illustrated in the corresponding forest plots.
Results
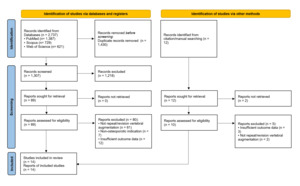
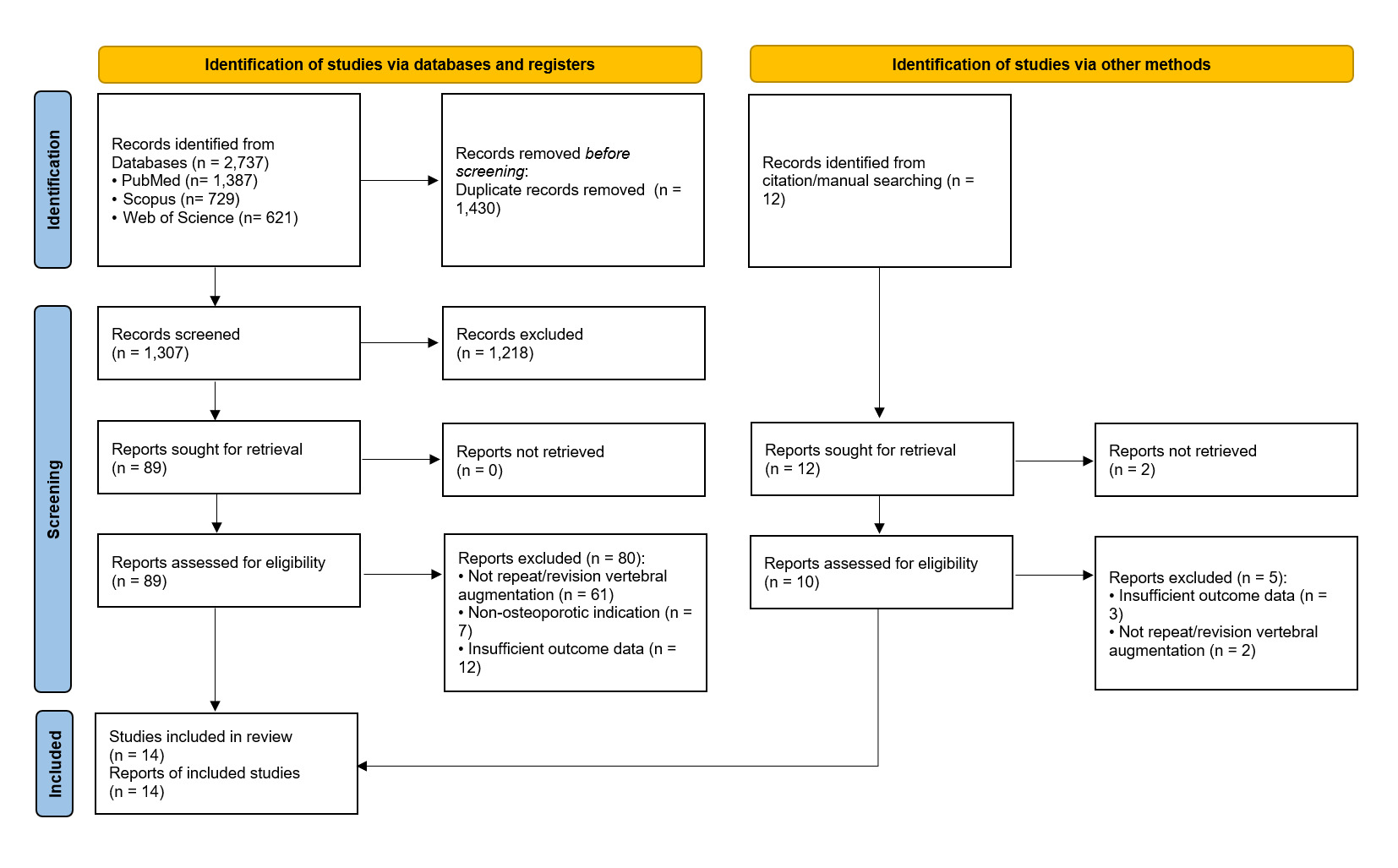
A total of 2,737 records were identified through database searches (PubMed, Scopus, and Web of Science), and an additional 12 were obtained through manual citation screening. After removal of 1,430 duplicates and application of the inclusion criteria, 14 studies were included in the review, which were all retrospective cohort studies. The study selection process is illustrated in the PRISMA flow diagram (Figure 1).
The 14 cohort studies, published between 2002 and 2025, included 320 patients and 358 treated vertebral levels undergoing repeat vertebral augmentation for recurrent or adjacent osteoporotic fractures. Sample sizes ranged from 6 to 50 patients per study. Across cohorts, 60 males and 261 females were represented. The mean patient age ranged from 65 to 80 years, with a pooled mean of approximately 73 years. Mean follow-up durations varied from 3 to 24 months, most commonly between 12 and 24 months. Reported intervals between the initial and repeat procedures ranged from 11 to 270 days, with both early and delayed reinterventions included. Most treated levels were located at the thoracolumbar junction (T11–L2), which accounted for approximately 73% of all vertebrae, followed by the upper thoracic and lower lumbar regions. Both PVP and PKP were represented among the included studies. Procedural combinations varied, including PVP-to-PVP, PKP-to-PKP, and cross-technique reinterventions, reflecting heterogeneity in clinical practice patterns and operator preference. All studies were retrospective in design, with most of the procedures being PVP and the more recent series including PKP. Baseline characteristics of the included studies are presented in Table 1.12–25
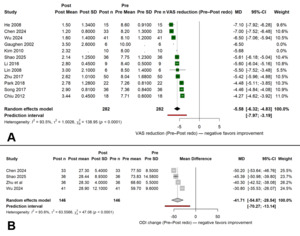
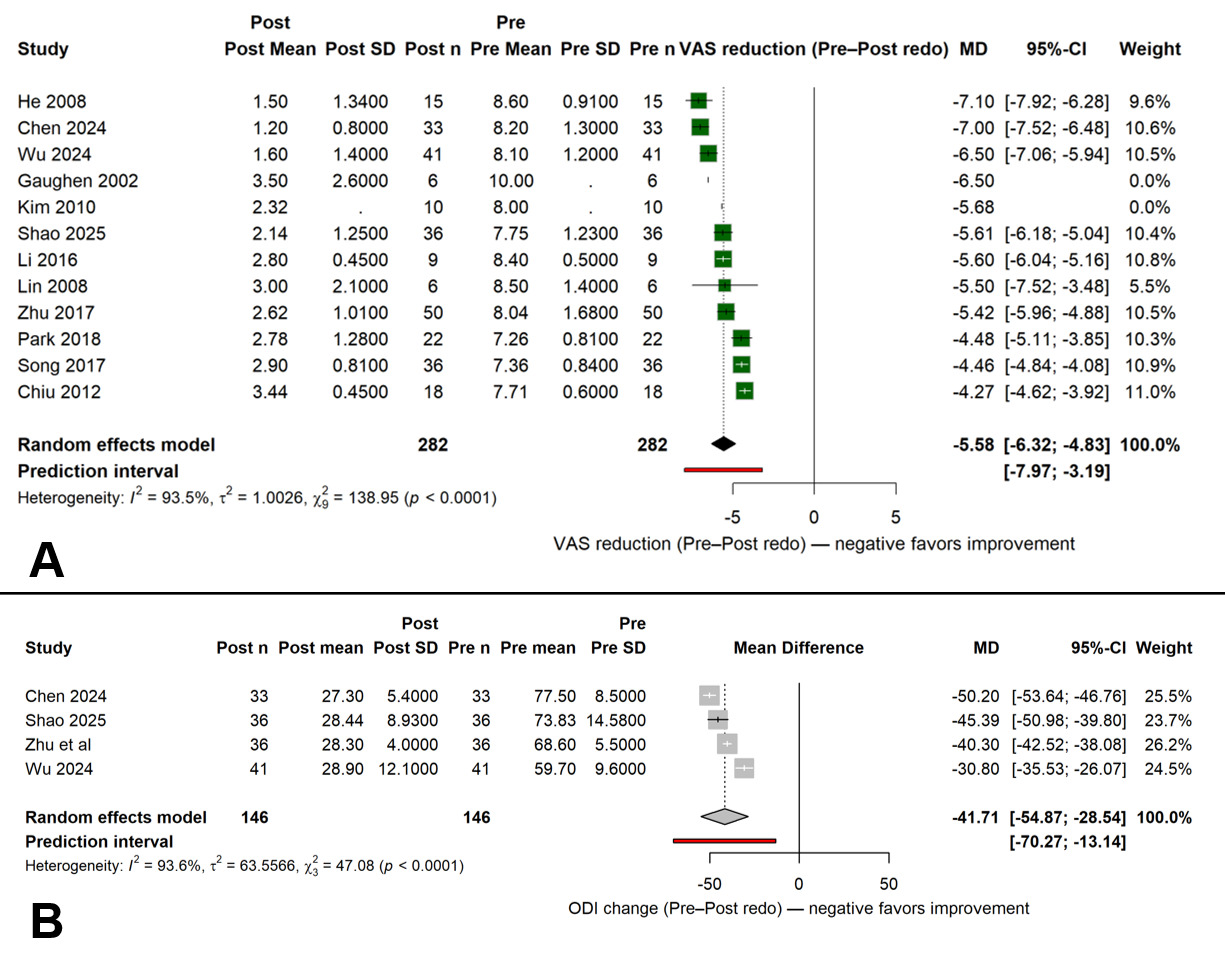
All studies reported postoperative improvement in pain scores. The pooled mean difference in VAS between pre- and post-procedure assessments was −5.58 points (95 % CI, −6.32 to −4.83; I² = 93.5 %; p < 0.001). The direction of effect was consistent across all cohorts, with every study showing a reduction in pain intensity following re-augmentation. Heterogeneity was mainly attributed to differences in baseline pain severity and follow-up duration. Four studies reported functional outcomes, yielding a pooled ODI change of −41.7 points (95 % CI, −54.9 to −28.5; I² = 93.6 %; p < 0.001). Despite the limited number of datasets, all studies showed postoperative functional improvement. Follow-up intervals for ODI assessment ranged from 3 to 24 months, and no study reported deterioration in functional capacity over time. The pooled pain and functional outcomes are illustrated in Figure 2.
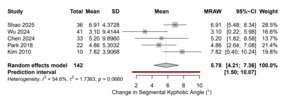
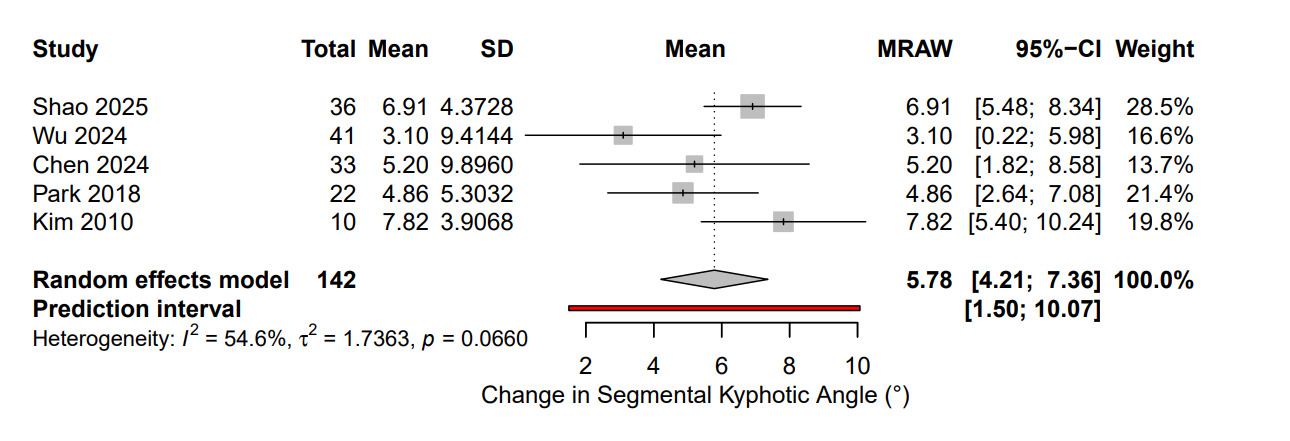
Radiological outcomes were available in five studies. The pooled correction in SKA was +5.78° (95 % CI, 4.21–7.36; I² = 54.6 %; p = 0.066). The pooled radiologic outcomes are shown in Figure 3. The angular correction values varied depending on the fracture level and technique used. A lot of data on vertebral height parameters were heterogeneous and often measured using different radiographic reference points. VHL, Mean anterior (AVH) and middle vertebral height (MVH) values were insufficient in number and consistency to do a quantitative synthesis.
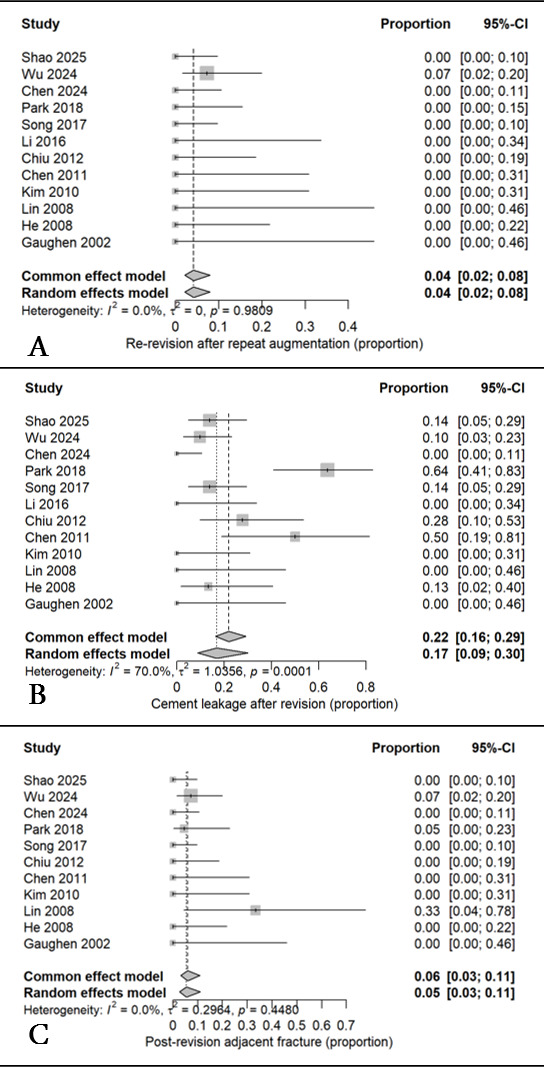
Complications were documented in all cohorts. The pooled rate of cement leakage was 17 % (95 % CI, 9–30 %; I² = 70 %), while adjacent vertebral fractures occurred in 5 % (95 % CI, 3–11 %; I² = 0 %). The pooled rate of re-revision was 4 % (95 % CI, 2–8 %; I² = 0 %). Leakage events were typically minor and detected radiographically without clinical symptoms. No cases of infection, cement embolism, or new neurological deficit were reported across any study. The pooled complication rates with low incidences of cement leakage, adjacent vertebral fracture, and re-revision are presented in Figure 4.
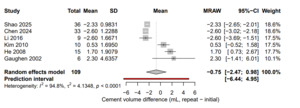
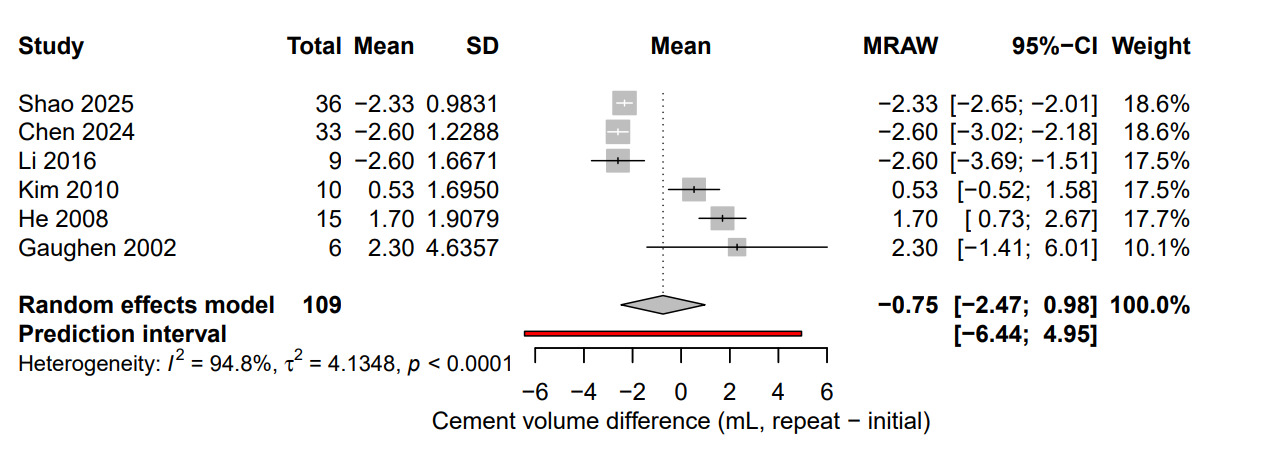
Procedural details were available in six studies comparing cement injection volumes between the initial and repeat augmentations. The pooled mean difference was −0.75 mL (95 % CI, −2.47 to 0.98; I² = 94.8 %; p = 0.23). Most studies reported similar cement volumes between procedures, and heterogeneity primarily reflected differences in cement type, viscosity, and injection protocol. The comparison of cement injection volumes between the initial and repeat procedures is illustrated in Figure 5. Bone density was reported in only a few of the included studies, using either T-scores or volumetric measurements in g/cm³. Mean T-scores ranged from −2.6 to −5.3, while reported bone density values ranged from 0.44 to 0.89 g/cm³. Because these data were available for only a small number of studies and reported using different metrics, they were not included in the pooled analysis.
Sensitivity analyses performed by excluding each study one by one did not change the pooled estimates significantly. Funnel plot inspection for VAS reduction did not demonstrate asymmetry, and Egger’s regression test showed no statistically significant publication bias (p = 0.42). Formal assessment for the other outcomes was not done due to the limited number of studies included in each analysis. A small number of articles mentioned a mix of unipedicular and bipedicular approaches, but the reporting was inconsistent and had no correlation with the analyzed outcomes. Therefore, this data was excluded from quantitative synthesis as well.
Risk of bias was generally low to moderate across the included studies. The main limitations related to potential confounding factors and selection bias, which is common in retrospective designs. None of the studies were judged to have a serious or critical risk of bias in any domain.
Discussion
To date, there is no previous meta-analysis that addressed repeat vertebral augmentation outcomes. This systematic review and meta-analysis is the first comprehensive synthesis on outcomes following repeat vertebral augmentation for recurrent or adjacent OVCFs. Across 14 retrospective cohorts including 320 patients and 358 treated vertebrae, repeat PVP or PKP consistently showed significant clinical improvement. The pooled mean reduction in pain intensity was −5.6 points on the VAS, accompanied by a mean functional improvement of −41.7 points in the ODI. Radiologic correction was modest but still significant, with a mean increase of +5.8° in SKA, a partial restoration of local alignment. Complication rates remained within the range reported for primary augmentation procedures, with cement leakage in 17%, adjacent fractures in 5%, and re-revision in 4% of cases. Even though it is a relatively small sample, the effects were uniform across all studies. These findings indicate that repeat vertebral augmentation remains an effective intervention for selected patients presenting with recurrent or adjacent osteoporotic fractures after initial augmentation.
Recurrent or adjacent vertebral fractures after augmentation are events that likely result from a combination of biological and mechanical factors rather than a single technical cause. The mean age across the included studies was around 73 years, including patients with advanced osteoporosis and poor bone quality. Older individuals naturally have lower bone density and deteriorating trabecular structure, both of which increase the risk of refracture even after successful initial augmentation. Most patients were female, which is consistent with the known predominance of postmenopausal osteoporosis. Recent meta-analyses support this pattern of fragility and mention that low bone mineral density, smoking, and diabetes are all associated with a higher risk of refracture.10,26 This overall secondary fracture incidence was reported to be around 17% during a median follow-up of 21 months.10,26 In our pooled analysis, adjacent vertebral fractures occurred in approximately 5% of patients, which is similar to the rates reported after primary augmentation. In a meta-analysis by Fan et al., no significant increase was found in adjacent-level fractures compared to the subgroup who got conservative treatment, suggesting that these events are more likely to be due to an underlying disease process.27 The pooled re-revision rate of 4% further supports the durability of repeat augmentation and indicates that once spinal stability is restored, recurrent failure at the same level is very uncommon.
On the other hand, follow-up duration was a factor that varied a lot and ranged from one month up to two years. This variability could explain part of the heterogeneity observed in the VAS and ODI results. Short-term follow-up studies tended to report greater immediate pain reduction, whereas those with longer observation periods captured more adjacent fractures and gradual loss of correction. These differences highlight the importance of considering follow-up time when interpreting pooled estimates.
Although cement volume is not a direct outcome measure, it was included in the analysis because it can influence both the risk of recurrence and the success of a repeat procedure. Earlier authors have suggested that incomplete cement filling during the first PVP might explain persistent pain.28,29 In a commentary on a study by Chiu et al., Wang et al. emphasized that repeat augmentation can only succeed when the surgeon can inject enough cement into the “responsible” area of the vertebral body.20,30 Our pooled analysis, however, found no significant difference in cement volume between the initial and repeat procedures (mean difference −0.75 mL, p = 0.23). This suggests that inadequate filling alone is unlikely to be a responsible factor for most cases of recurrent pain or collapse.
Bone quality appears to be a far more important factor. The few included studies that reported bone-density data described severely reduced T-scores (−2.6 to −5.3) and low volumetric values (0.44 to 0.89 g/cm³), which suggest advanced systemic osteoporosis. Poor bone stock weakens the trabecular scaffold surrounding the cemented level and increases local stress at adjacent endplates, causing new fractures of the spine regardless of the cement amount used.31 Taken together, these findings support the idea that repeat fractures after vertebral augmentation arise primarily from ongoing skeletal fragility and altered load transfer rather than technical inadequacy at the time of the initial procedure.32
Radiologic outcomes were less consistent than the clinical measures but still provided useful insight into the mechanical effectiveness of repeat vertebral augmentation. The pooled correction in SKA across five studies was +5.8°, which is a modest yet measurable restoration of vertebral alignment. This degree of angular correction is smaller than that typically reported after primary vertebral augmentation, where mean SKA improvements often exceed 7°–10°.33,34 In the current synthesis, the study by Shao et al. was the only one included in the SKA pooled analysis that involved PKP, which means that slight angular improvement can also occur following PVP.12 This is likely the result of partial vertebral height restoration due to the prone positioning of the patients, internal hydraulic pressure during cement injection, and re-establishment of anterior column support. In addition, by the time a repeat augmentation is performed, the treated vertebra already contains hardened cement, which limits further height recovery or balloon expansion.28,35
Data on vertebral height parameters such as anterior, middle, or posterior vertebral height were reported too inconsistently to include them in the quantitative pooling. Many studies used different measurement reference points or failed to distinguish between the previously cemented and newly fractured levels. In those that did, results generally followed the same trend as SKA, showing partial recovery of height without complete correction. This highlights the need for more standardized radiographic assessment in future studies.
Across all included studies, repeat vertebral augmentation was associated with a low incidence of major complications and an overall safety profile comparable to that of primary PVP or PKP. The pooled rate of cement leakage was 17%, which falls within the 10–25% range commonly reported after primary augmentation.36 Importantly, nearly all leakage events were minor and radiographically detected without neurological or pulmonary consequences. No study described symptomatic embolism, infection, or new neurological deficits. Adjacent vertebral fractures occurred in approximately 5% of patients, which again is comparable to rates observed following primary procedures.37 The pooled re-revision rate of 4% further supports the procedural durability of repeat augmentation and that once adequate stability is restored, the likelihood of failure again level remains low.
Taken together, these findings demonstrate that performing a repeat PVP or PKP does not appear to increase the risk of cement-related or mechanical complications. The technical complexity may be slightly greater but the safety outcomes reported across the literature reinforce that re-augmentation is a low-morbidity option in the appropriately selected patients.
This review has several important strengths. It is, to our knowledge, the first meta-analysis to quantitatively synthesize clinical, radiologic, and procedural outcomes of repeat vertebral augmentation. The analysis incorporated data from more than 300 patients and represents one of the largest cohorts to date in this rare clinical scenario. By including cement volume and bone density, this study also provides new insight into the biomechanical and systemic factors that may underlie recurrent vertebral collapse and compare it to the outcomes. In addition, by using the PRISMA methodology, prospective PROSPERO registration, and consistent statistical approaches adds to the transparency and reproducibility of the results.
Nonetheless, several limitations should be recognized. All included studies were retrospective case series or cohorts, so they were more likely to be subject to selection bias, incomplete reporting, and unmeasured confounding. The total sample size remains relatively small, and most studies lacked control groups for comparison with primary procedures. Substantial heterogeneity was observed for several outcomes which caused differences in follow-up duration, baseline pain severity, and treatment protocols. Radiological and functional measures were also inconsistently reported. Finally, although demographic variables such as age, sex distribution, and bone density were extracted and analyzed descriptively, the limited number of studies and the lack of individual participant data made formal meta-regression not possible. These limitations underscore the need for larger, prospective, and methodologically uniform studies to validate and refine these findings.
It is also important to note that 13 of the 14 included studies were conducted in East Asian countries (China, Taiwan, and South Korea), with only one study originating from the United States. Factors such as differences in bone density, body habitus, vertebral morphology, and surgical technique may influence both outcomes and complication rates. In addition, a Chinese study reported an increase in recognized vertebral fractures among adults aged ≥ 50 years from approximately 85 to 152 per 100 000 person-years between 2013 and 2017.38 Similarly, a Taiwanese nationwide cohort study including more than 24,000 older adults with an initial osteoporotic fracture reported that approximately 45% sustained a repeat fracture within one year, corresponding to an incidence rate of 950.5 per 1,000 person-years during the first year of follow-up.39 This results in vertebral augmentations being performed more frequently and often earlier in East Asia compared with Western countries. As a result, the findings of this meta-analysis should be interpreted primarily within that demographic and healthcare context, and further studies from Western and multiethnic populations are needed to confirm the generalizability of these results.
The findings of this meta-analysis support repeat vertebral augmentation as a practical and effective treatment for carefully selected patients who experience recurrent pain after initial PVP or PKP. When imaging confirms a new or progressive collapse at a cemented or adjacent vertebra and a safe access route exists, re-augmentation can provide meaningful symptom relief with low procedural risk. Although cement volume and distribution remain important technical considerations, this analysis suggests that systemic osteoporosis and altered spinal biomechanics are the principal contributors to recurrence, rather than inadequate cement filling alone. Addressing underlying bone fragility through medical management and fall-prevention strategies should therefore accompany any interventional approach.
From a broader perspective, these results reinforce the role of vertebral augmentation not only as an acute pain-relieving procedure but also as a reproducible option when failures occur. Future research should focus on prospective, multicenter registries with standardized radiologic measurements, uniform definitions of recurrence, and long-term follow-up to better characterize the durability of re-augmentation. Integrating bone health optimization and biomechanical assessment into treatment algorithms may ultimately reduce the need for repeat procedures.
Conclusion
In conclusion, repeat PVP and PKP offer substantial and reliable pain reduction, functional improvement, and radiographic stabilization in patients with recurrent or adjacent OVCFs. The complication rates are low and similar to primary augmentation. Recurrence after initial treatment appears to stem more from progressive bone fragility than from procedural shortcomings, which shows the importance of osteoporosis management besides surgical intervention.
Acknowledgements
None
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Stylianos Kapetanakis, Mikail Chatzivasiliadis, Nikolaos Gkantsinikoudis, Constantinos Chaniotakis, Panagiotis Konstantinou, Lazaros Kostretzis, Krikor Gkoumousian, Iosafat Pinto, Paschalis Tsioulas, and Konstantinos Ditsios. The first draft of the manuscript was written by Mikail Chatzivasiliadis, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Competing Interests
The authors have no relevant financial or non-financial interests to disclose.
Informed Consent
Written informed consent was obtained from the patient and the parents for publication of this study and any accompanying images.