1. Introduction
Osteoarthritis (OA) is categorized as a chronic and progressive degenerative disease and is one of the leading causes of disability. This form of arthritis is commonly known to cause considerable pains, limiting mobility and functional capability within specific body joints in those affected.1,2 It is considered one of the most common causes for joint pain and disability, while often also regarded as an age-related condition due to the increased occurrence of OA in patients that are 45 years old and above.3 One common form of OA that has affected multitudes through said factors is knee osteoarthritis (KOA). Characterization of knee OA is generally identified through the degeneration activity of the joint, causing loss of cartilage in the site of injury. This can be also accompanied by a damaged synovial membrane, as well as other potential joint components within the injury site.4
Biochemically, OA is characterized by the upregulation of pro-inflammatory cytokines such as IL-1β, TNF-α, IL-6, IL-17, and IL-18.5–7 In addition to cytokines, matrix metalloproteinases (MMP), a family of zinc-dependent enzymes, are known for their ability to degrade ECM of the articular cartilage, which mainly consists of collagens and proteoglycans.6 MMP-1, a known collagenase, is frequently found to be elevated in osteoarthritis,8,9 while MMP-2 and MMP-9 are responsible to cleave ECM, cytokines, and chemokines, thus enhancing their activities.10 The activation of MMPs in the cartilage degeneration process affects the stability of cartilage oligomeric matrix protein (COMP) levels in the synovium. COMP plays a crucial role in the secretion process of fibrillogenesis and can interact with various components of the extracellular matrix (ECM), such as collagen types I, II, and IX, fibronectin, and others.11
The main focus of OA treatment is not only to reduce pain but also to improve function and quality of life. Treatment strategies for OA are designed to relieve pain, correct structural abnormalities, and restore joint function. Various approaches have been employed, such as oral nonsteroidal anti-inflammatory drugs (NSAIDs), physical therapy, and intra-articular injections.12 Other agents, including corticosteroids (CS), platelet-rich plasma (PRP), and clodronate have demonstrated anti-inflammatory and analgesic effects, yet their limited biological activity restricts long-term benefits.13,14 In more severe cases, surgical procedures such as total knee replacement (TKR), arthroscopy, osteotomy, and arthroplasty are considered. While effective, these interventions carry drawbacks: TKR is associated with morbidity and limited implant lifespan, osteotomy risks infection and failure, and arthroscopy yields variable outcomes depending on the technique used (lavage, debridement, abrasion arthroplasty).15,16 Arthroscopy, however, offers the advantage of being applicable in earlier disease stages, where it may improve joint mobility and reduce pain by removing debris, inflammatory factors, or unstable chondral flaps.17–19
Recently, in-depth research in regenerative medicine, particularly with mesenchymal stem cells (MSCs) and their secretomes, have introduced promising therapeutic strategies for OA.20 MSC injections offer excellent histocompatibility and strong bioactivity, supporting cartilage repair and regeneration through both direct cell activity and the secretion of extracellular matrix (ECM) components.21 Compared with MSCs, their secretome demonstrates greater stability and exerts multiple therapeutic effects, including anti-inflammatory, immunomodulatory, pro-angiogenic, and anti-apoptotic actions. The secretome also has the advantage of containing various growth factors, exosomes, and microvesicles, which contribute to inhibiting inflammation and apoptosis, as well as promoting neurogenesis, angiogenesis, synaptogenesis, and other regenerative processes.22
For these reasons, this study aims to evaluate and compare the therapeutic effectiveness of UC-MSCs and UC-MSC secretomes in patients with and without arthroscopy, as well as to investigate their effects on biomarker expression in osteoarthritic synovial fluid mesenchymal stem cells (SFMSCs) at the laboratory scale. We hypothesize that combining arthroscopy with UC-MSCs or their secretomes may enhance cartilage repair, improve clinical outcomes, and modulate biomarkers in knee OA.
2. Materials and Methods
The study had a randomised, open-labelled randomized clinical trial, and it fulfilled the specified inclusion criteria. The research was done at the Gatot Soebroto Army Hospital in Jakarta, Indonesia. From January 2020 to December 2023. The study was conducted in accordance with the guidelines outlined in the Declaration of Helsinki by the researchers. The study (No.41/VIII/KEPK/2023) received permission from the Ethics Committee of Gatot Soebroto Army Hospital. The research protocol identified as NCT04314661, was properly submitted to the Clinical Trials Registry.
2.1. Study Participants/Patient Selection
All participants provided written informed consent, and the study received approval from the ethics committee. Following that, patients with knee osteoarthritis were recruited. Inclusion and exclusion criteria were applied during the recruitment procedure and thus, resulting in seven female patients that have successfully passed and are eligible for the study. The following inclusion criteria for the study: ages 55-70, patients diagnosed with knee OA grade 2-3 identified by two different observers based on Kellgren-Lawrence scale, no common or local infection activity, and finally an insignificant change on patients’ examination of both biochemistry and haematology scans requiring contraindication. Patients admitted must also have understood the nature of the study and properly acknowledged every detail within the informed consent. Exclusion criteria included: signs of infection or positive serology for HIV, hepatitis, and syphilis; a history of cancer within the patient’s family or themselves and tumour marker value evaluation greatly exceeds standard range; a genetic disease that causes significant abnormal morphology which can affect cell transplantation and result interpretation; a three month prior of intra-articular drug injection on the knee; participated in a clinical trial 30 days prior of study; signs of other medical conditions that can affect patients health, and finally patient is a subordinate or low-ranking member (Hospital requirement). Subjective and Objective parameters were also conducted in this study; with Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) and visual analogue scale (VAS) as the subjective parameters; and enzymatic activity examination of COMP, MMP-13, and IL-6 from knee fluid, as well as cartilage growth check using MRI T2 Mapping as the objective parameters. We enrolled seven patients (F = 6, mean age: 56) with knee OA classified as stage 1-3 according to Kallgren and Lawrence classification.
2.2. Sample Collection and Preparation
Beforehand, available umbilical cord candidate donors were referenced and provided by a partner medical institution known as PT. Prodia StemCell Indonesia. The donor candidate chosen must fulfil the criteria of 20-25 years old, no cancer history, and result in negative examination of HIV, hepatitis b and c, CMV, syphilis, toxoplasma, rubella, and herpes. If the donor is eligible, informed consent is given to the donor for acceptance. Following approval, the donor umbilical cord is harvested during childbirth around 5-10 cm by the specialized doctor. The umbilical cord is then washed in normal saline to clear excess blood and proceed to be delivered to PT. Prodia StemCell Indonesia GMP Laboratory for processing into UC-MSC and secretome.
2.3. Randomisation and Intervention
The participants who fulfilled the predetermined criteria for inclusion and exclusion were allocated into two groups using a randomisation procedure. The allocation of subjects into two groups was achieved using block randomisation in a 1:1 ratio, following the acquisition of their informed consent. The randomisation process was conducted using a random number generator accessible via the following link: https://stattrek.com/statistics/random-number-generator.aspx (accessed on 1 December 2023).
Pre-conditioned groups are paired with two different groups, one with arthroscopy, and the other without arthroscopy. Each group received an intra-articular injection consisting of an initial 2 mL dose of secretome, followed by 10 million UC-MSCs, and additional 2 mL doses of secretome administered biweekly. Patients also carry out a follow-up evaluation for every 6- and 12-months following stem cell injection.
2.4. Biochemical Assay
Synovial fluid samples were collected from each participant at baseline and at 12 weeks post-intervention, with an approximate volume of 5 mL per collection. Samples were processed by centrifugation to obtain the supernatant, which was then analyzed using a pooling strategy for inflammatory and matrix-degrading markers. Concentrations of interleukins (ILs), interferon-gamma (IFN-γ), cartilage oligomeric matrix protein (COMP), and matrix metalloproteinases (MMPs) were quantified using the following assays:
-
Interleukins (ILs): LEGENDplex™ Human Inflammation Panel 1 (13-plex; Cat. No. 740808, BIOLEGEND), including IL-1β, IFN-γ, MCP-1, IL-6, IL-8, IL-12p70, IL-17A, IL-18, and IL-23.
-
Matrix Metalloproteinases (MMPs): Human MMP Premixed Magnetic Luminex Performance Assay (4-plex; Cat. No. FCSTM07-04, Luminex R&D), including MMP1, MMP3, MMP7, and MMP13.
-
Cartilage Oligomeric Matrix Protein (COMP): ELISA Kit (Cat. No. RD194080200), used for quantification of COMP levels.
2.5. Arthroscopic Procedure
Pre-arthroscopy, each patient underwent multiple examinations such as a laboratory test for: complete blood check-up, heart function test, liver function test, and blood coagulation; X ray thoric test for lung function test; and finally, surgery tolerance from specialized doctor of internist, pulmonologist, cardiologist, and anaesthesiologist. After the examination, eligible patients underwent arthroscopy. Patients were first administered spinal anaesthesia, then two small incisions of 1 cm in size are made below the kneecap, into the outside and inside (anterolateral portal and anteromedial portal) layer. Both arthroscopic camera and work tool was inserted into the incision through anterolateral portal and anteromedial portal, respectively, to provide view. Sterile fluid was inserted into the knee joint, followed by a diagnosis of inflammation (synovitis), cartilage damage, and osteophyte presence. The knee joint diagnosed factors were cleansed, as well as the removal of dirt and damaged tissue. After cleansing, the knee joint was washed with sterile fluid added with antibiotics (gentamicin 80), and fluid was drained from the joint. Closing, the surgical wound was sutured with single knot sutures.
3. Results
This study evaluated the therapeutic effects of UC-MSCs and UC-MSC-derived secretome in OA patients through both clinical outcomes and synovial fluid (SF) analyses.
3.1. Clinical Outcomes
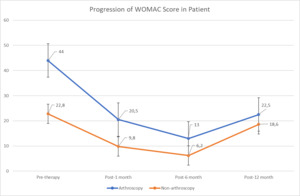
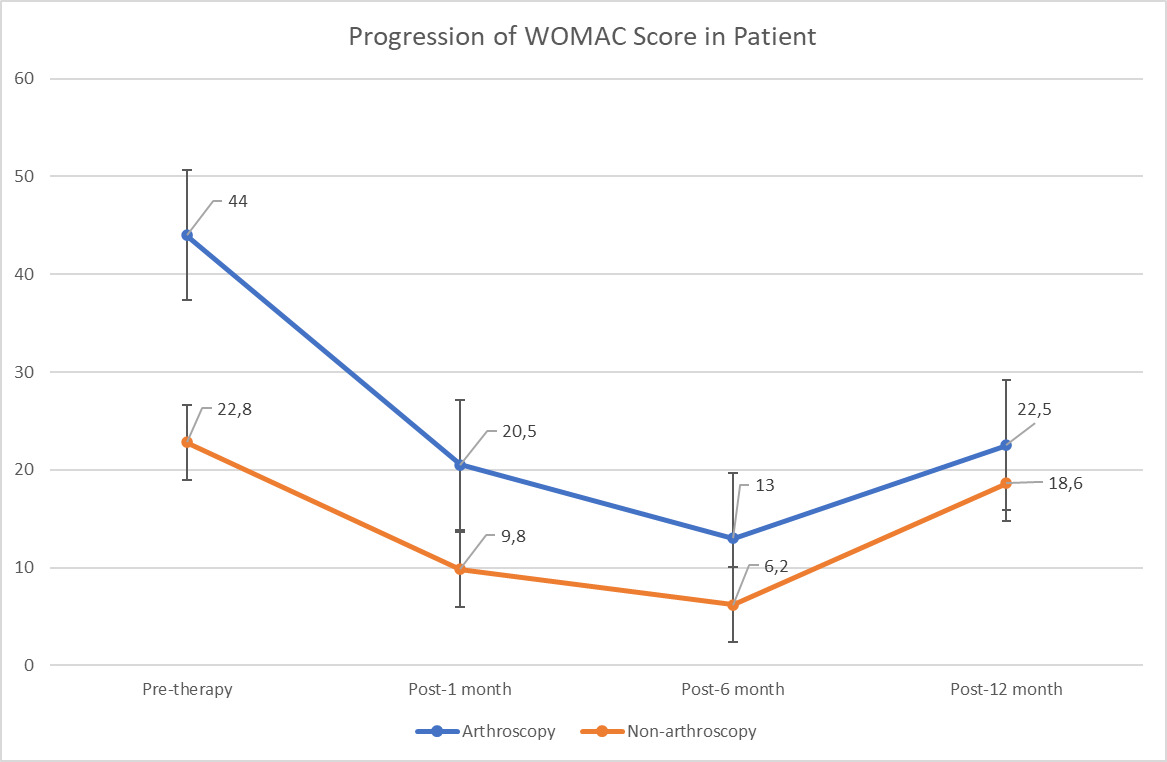
Clinical evaluations following UC-MSC and UC-MSC secretome treatment were performed using both subjective and objective measures. Patients were all female and coded based on initials (Table 1). Two patients underwent arthroscopy (Da and E), while five patients did not (I, T, C, R, and De). Treatment was administered in three escalating doses, and follow-up assessments were conducted at baseline, 1 month, 6 months, and 12 months. Outcomes were measured using the WOMAC and VAS scores.
Both treatment groups demonstrated reductions in WOMAC scores, indicating improved functional outcomes. VAS scores were excluded due to inconsistent reporting and missing data; therefore, WOMAC scores were used as the primary outcome. Patients who underwent arthroscopy showed initial WOMAC values of 40 and 48, which improved to 9 and 32 at 1 month, further decreased to 2 and 24 at 6 months, but partially worsened to 10 and 35 at 12 months. In the non-arthroscopy group, baseline WOMAC values ranged from 15 to 32, with consistent reductions across all patients at 1 month (scores 0–12) and 6 months (0–18). By 12 months, four of the five patients demonstrated sustained or improved functional recovery, with WOMAC scores ranging from 0 to 53.
Figure 1 illustrates the overall trajectory of WOMAC score progression in both groups. The greatest functional improvement occurred at 6 months post-treatment, with sustained benefit in most patients at 12 months. Notably, two arthroscopy patients (Da and E) and one non-arthroscopy patient (T) experienced partial loss of improvement at the final follow-up. Based on WOMAC total score interpretation (0 = none; 1–23 = mild; 24–48 = moderate; 49–72 = severe; 73–96 = extreme), most patients transitioned from “mild–moderate” impairment at baseline to “none–mild” impairment by 6 months. At 12 months, however, two patients (Da and E) reverted to the moderate range, and one patient (C) remained in the mild range.
3.2 Biochemical Results
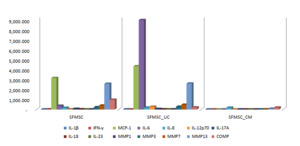
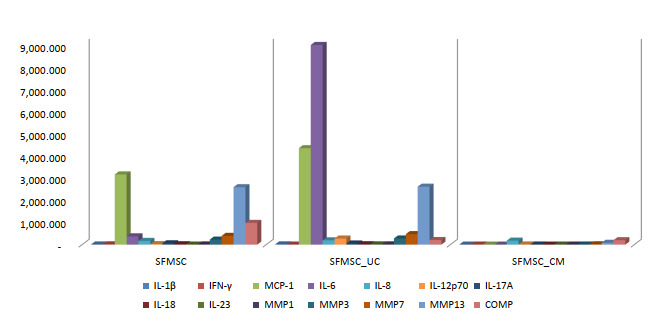
To investigate the underlying mechanisms, synovial fluid (SF) from OA patients was analyzed following co-culture with UC-MSCs and their secretome. Co-culture of OA SF with UC-MSCs resulted in significant increases in interleukin-6 (IL-6) and interleukin-12p70 (IL-12p70), accompanied by reductions in interferon-gamma (IFN-γ) and COMP compared to SF cultured alone. Other inflammatory cytokines and matrix-degrading enzymes—including interleukin-1 beta (IL-1β), monocyte chemoattractant protein-1 (MCP-1), interleukin-8 (IL-8), interleukin-17A (IL-17A), interleukin-18 (IL-18), interleukin-23 (IL-23), and matrix metalloproteinases (MMP-1, MMP-3, MMP-7, MMP-13)—did not show significant changes. These findings suggest that UC-MSCs selectively modulate key inflammatory and cartilage-degrading factors.
In contrast, supplementation with UC-MSC secretome produced broader immunomodulatory effects, with significant reductions in IL-1β, IFN-γ, IL-6, IL-12p70, IL-17A, IL-18, MMP-1, MMP-7, MMP-13, and COMP, while MCP-1, IL-8, IL-23, and MMP-3 levels remained unchanged. These in vitro results align with the clinical improvements observed, indicating that the secretome exerts a more comprehensive anti-inflammatory and anti-catabolic effect. Collectively, these findings support the therapeutic potential of UC-MSCs and their secretome in OA by mediating targeted immunomodulation and protecting cartilage integrity.
4. Discussion
The treatment application of using MSCs in OA patients was constantly proved to produce significant improvements in functional capability of patients in their quality of life. Qualitative measurements such as using WOMAC, and VAS scores all show improved values when evaluated after the treatment method of using MSCs. In our work however, there are some instances of WOMAC scores increasing in patients during the 12-month of observation post stem cell injections. This is also evident in other clinical article works such as in Emadedin et al. (2012) in which WOMAC index scores increased slightly during post 12-month stem cell injections as opposed to continually decreasing.4
Two patients in this study underwent arthroscopy before UC-MSC administration. Overall, patients who underwent arthroscopy demonstrated a greater reduction in WOMAC scores compared with those managed without the procedure. Theoretically, arthroscopy is expected to remove intra-articular debris and reduce pro-inflammatory cytokines that contribute to synovitis.3,23,24 By clearing these inflammatory components and improving the joint microenvironment, the therapeutic effects of stem cells and their secretome may be enhanced, allowing them to function more effectively in promoting tissue repair and symptom improvement.
Interestingly, our findings highlight UC-MSCs selectively modulate key inflammatory and cartilage-degrading factors. An inflammatory microenvironment prompts MSCs to secrete pro-inflammatory mediators such as IFN-γ as part of their initial response. This upregulated secretion supports the recruitment of surrounding endogenous cells within the synovium, initiates angiogenic processes, and facilitates the homing of these cells to the injured site. Together, these actions contribute to subsequent regenerative activity aimed at restoring homeostasis within the damaged synovium.25–27 It was mentioned before that the activation of MMPs in the cartilage degeneration process affects the stability of COMP levels in the synovium, this is in line with our findings where COMP results decreased, and inversely proportional to the number of MMPs which remained high. Consistent with Pourgholaminejad et al. (2016), and Croes et al. (2015), pro-inflammatory also enhances the multilineage differentiation potential and immunomodulatory capacity of MSCs, suggesting that this initial “priming” phase is not irritative but may instead potentiate their therapeutic function in osteoarthritic joints.25,28
The culture medium that contains secretome, the set of molecules released by stem cells, is referred to as conditioned medium (CM)29-30. In contrast to the concentration of biomarkers co-cultured with UC-MSCs, SF-MSCs co-cultured with CM actually showed a significant decrease in the levels of interleukins, MMPs, and COMP as shown in the result. This is consistent with previous reports demonstrating that MSC-derived secretomes possess a distinct molecular profile compared to other secretomes. Specifically, they are enriched with angiogenic factors, contain lower levels of MMPs, and exhibit high production of TGF-β, chemokines, and anti-inflammatory cytokines. Such a composition makes the UC-MSC secretome a potent driver of inflammation regulation, which may explain the biomarker reduction observed in our findings. Another study by Lee et al., (2024) stated that SF-UC-MSC conditioned media effectively reduced IL-6 levels, a proinflammatory biomarker. Lee also mentioned that SF-UC-MSC conditioned media downregulates COX-2 gene expression, an enzyme that promotes inflammation.29
In conclusion, the clinical and in vitro findings indicate that UC-MSCs and their secretome exert therapeutic effects through a coordinated set of mechanisms. Arthroscopy serves as the initial step by removing intra-articular debris and inflammatory by-products, thereby improving the joint microenvironment and allowing subsequent biological therapies to act more effectively. Following this mechanical clearing, the UC-MSC–derived conditioned medium provides an early anti-inflammatory influence by rapidly downregulating pro-inflammatory cytokines and MMPs. Finally, UC-MSCs delivered thereafter promote proliferation, differentiation, and additional cytokine suppression.
This study is limited by a small sample size, short follow-up duration, and absence of imaging-based structural assessment, as well as the missing of VAS scores. The heterogeneity of patient severity and the use of arthroscopy in a subset also introduce variability. Future research should incorporate larger controlled trials, stratify patients by OA grade, and perform molecular profiling to identify responders and non-responders.
5. Conclusion
The combined administration of UC-MSC secretome and UC-MSCs, preceded by arthroscopic debridement, provides complementary therapeutic benefits in knee osteoarthritis. Secretome delivery rapidly reduces inflammation by suppressing pro-inflammatory cytokines and matrix-degrading enzymes, while UC-MSCs subsequently promote cartilage repair through enhanced proliferation and differentiation. Clinical improvements up to six months, supported by in vitro findings, highlight this two-stage approach as a promising strategy for both inflammatory control and joint regeneration in OA management.
Author Contributions
Conceptualization, Y.Y; K.L.D; C.R.S.; methodology, Y.Y; K.L.D; C.R.S; D.K.D.; validation, K.L.D; C.R.S.; formal analysis, Y.Y; K.L.D; C.R.S.; data curation, Y.Y; K.L.D; C.R.S; D.K.D; R.H.; writing—original draft preparation, Y.Y; K.K; D.K.D; R.H; L.M.U.; writing—review and editing, Y.Y; D.K.D; K.K; R.H.; visualization, Y.Y; D.K.D.; supervision, K.L.D; C.R.S.; project administration, Y.Y; R.H; D.K.D; K.K.; funding acquisition, C.R.S;. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by PT. Prodia StemCell Indonesia.
Institutional Review Board Statement
Not Applicable.
Informed Consent Statement
Written informed consent was obtained from all participants prior to study enrollment.
Data Availability Statement
Not Applicable.
Conflicts of Interest
The authors declare no conflict of interest.