Introduction
Total Joint Arthroplasty of the hip (THA) and knee (TKA) are among the most successful surgical procedures worldwide, delivering significant improvements in pain relief, mobility and quality of life.1,2 However, the success of TJA is undermined by periprosthetic joint infection (PJI), which is now the leading cause of revision in many registries, accounting for up to 27.2% and 25.7% revisions in THA and TKA respectively. Prevention remains paramount, as PJI treatment carries high morbidity, mortality and cost burden.3
The devastating consequences of PJI extends beyond surgical morbidity. It is associated with mortality rates of 11%, prolonged hospitalisation and profound psychosocial effects.4 Patients suffer reduced quality of life, loss of independence and 58% of patients report depressive symptoms throughout treatment and 21.9% continue to have symptoms at 5 years following PJI eradication.5 Economically, PJI management is 4.5–5 times more costly than uncomplicated TJA, with global healthcare systems projected to face expenditures exceeding $1.8 billion annually by 2030.6
Risk factors for PJI can be divided into modifiable and non-modifiable categories.7 Non-modifiable factors include age, sex, comorbidity burden and genetic predisposition.7 Modifiable factors include obesity, HIV, smoking, malnutrition, diabetes, operative time, perioperative traffic and air quality control.8 Importantly, airborne contamination is a modifiable factor that can be actively managed through ventilation strategies, behavioural interventions and rigorous perioperative protocols.
Methods
A review of the current literature was conducted using PubMed, MEDLINE, Scopus, and Google Scholar databases from inception to June 2025. Search terms included combinations of “air quality,” “laminar airflow,” “turbulent ventilation,” “operating theatre,” “arthroplasty,” “periprosthetic joint infection,” and “surgical site infection.” Relevant systematic reviews, registry analyses, experimental studies, and guidelines were considered. Additional sources were identified through manual screening of bibliographies. Articles not published in English or not directly addressing orthopaedic operating theatre air quality were excluded. Evidence was synthesised thematically to provide a comprehensive overview of environmental controls, human behavioural factors, surgical helmet systems, and regulatory or economic considerations in infection prevention.
Role of airborne contamination in PJI
The role of airborne contamination in PJI has been recognized for decades.9 Skin squames, respiratory droplets and staff movement all contribute to the microbial load in the operating room (OR), with particles ≥5 µm often acting as carriers of bacteria.10–12 Consequently, theatre design has evolved toward advanced ventilation systems, particularly laminar airflow (LAF), though recent meta-analyses and registry data repeatedly challenge its benefits.13–15
This review synthesizes evidence across five domains: (1) airborne contamination and ventilation strategies; (2) human behaviour and environmental interaction; (3) surgical helmet systems; (4) integration with patient and procedural risk factors and (5) economic and regulatory frameworks.
Airborne Contamination in the Arthroplasty OR
Sources of Airborne Microbe-Carrying Particles (MCPs)
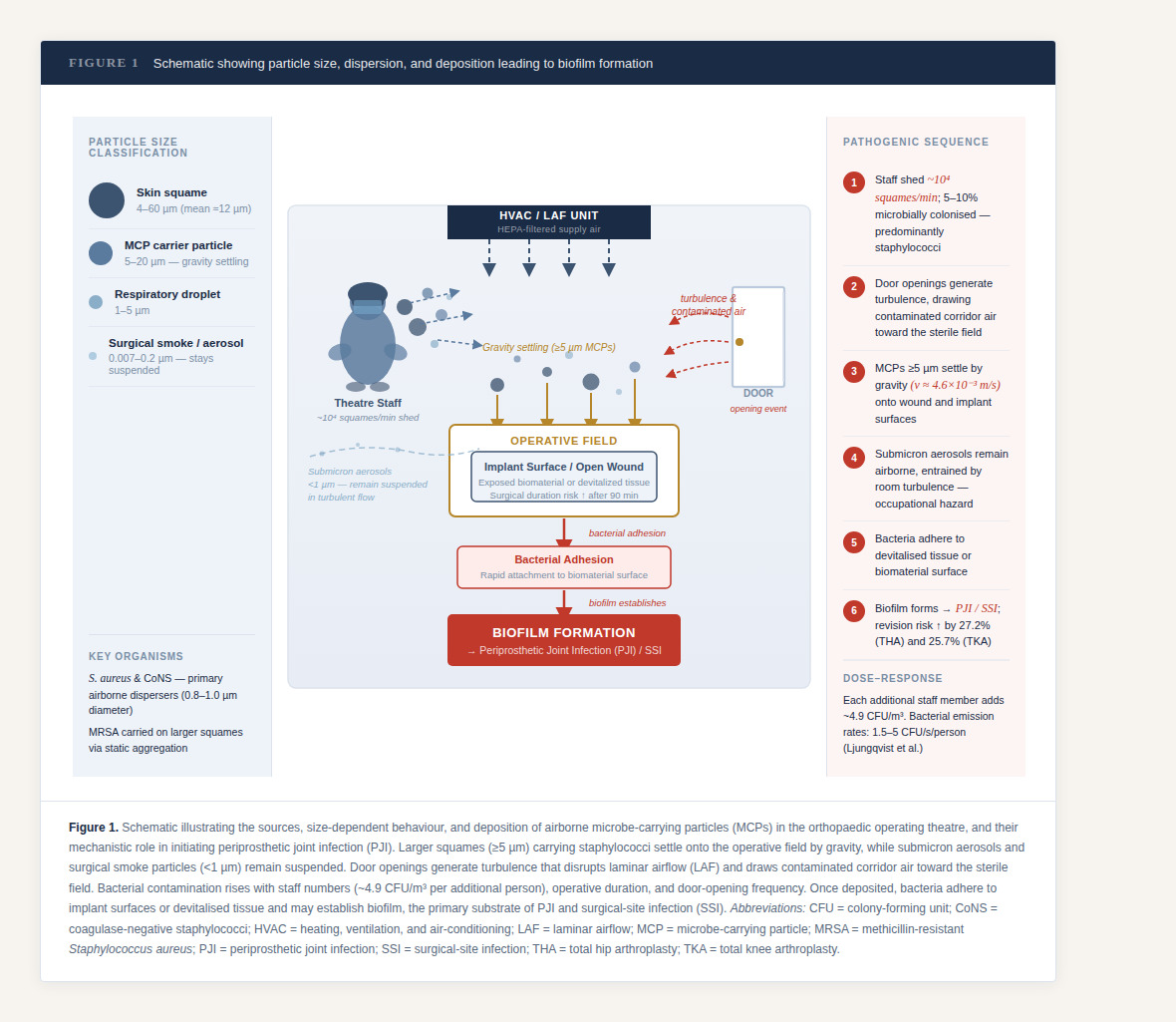
In the OR, airborne microbe-carrying particles (MCPs) arise chiefly from human desquamation and staff movement, augmented by handling of textiles/drapes and instrument activity. MCPs are typically shed as skin squames that measure ~4–60 µm (mean ≈12 µm) and frequently carry staphylococci from the skin microbiome.16–18
Particle Size, Transport, and Deposition
During moderate activity a single person may release ~10⁴ skin scales per minute, ~5–10% of which are microbially colonized.19 Particle size determines transport and deposition: bacteria-carrying squames in the ~5–20 µm range settle rapidly by gravity with a measured deposition velocity around 4.6 × 10⁻³ m·s⁻¹, contaminating the wound, instruments and implant surfaces, whereas submicron aerosols remain suspended and are entrained by room turbulence.20,21 Electrosurgery/laser “surgical smoke” adds vast numbers of ultrafine particles (down to ~0.007–0.2 µm), which, although primarily an occupational hazard, increase airborne particle burden in the sterile field and challenge ventilation/filtration performance22,23
Impact of OR Traffic and Door Openings
OR traffic and door openings disrupt airflow, draw in contaminated corridor air and are consistently associated with higher airborne and surface viable counts near the operative field.24,25 A dose–response relationship exists between airborne bacterial concentration and surgical-site sepsis, and landmark randomized work in TJA has shown that ultraclean (unidirectional) airflow and body-exhaust suits halved deep joint sepsis after THA/TKA compared with conventional ventilation.10,26 However, contemporary analyses note mixed PJI benefits for LAF in the context of modern antibiotic/bundle prophylaxis.27 Ventilation can lower airborne microbial counts; however, staff traffic consistently elevates particles and CFU near the field; registry data show no clear PJI reduction from LAF alone, strengthening the argument that traffic control is a key modifiable driver within PJI prevention bundles.28,29
Mechanism of Infection and Biofilm Formation
Mechanistically, once bacteria deposit onto devitalized tissue or newly implanted biomaterials with rapid adhesion may cause biofilm formation potentially driving ensuing PJI and SSI.30,31
Evolution of Theatre Ventilation Systems
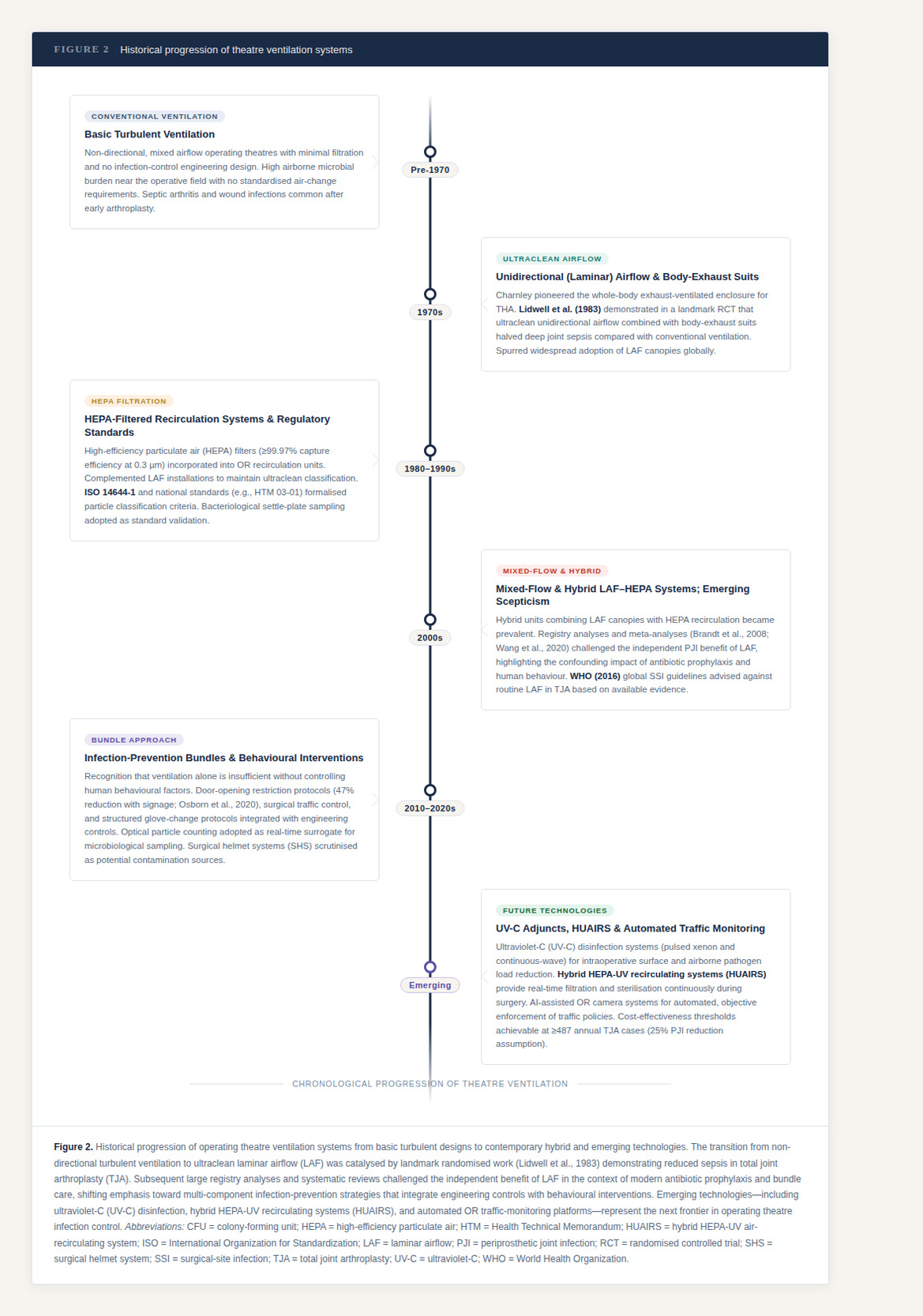
Maintaining a sterile environment during TJA is central to reducing PJI risk. ORs have evolved from basic turbulent ventilation systems to ultraclean LAF environments. In the 1970s, Lidwell et al.10 demonstrated reduced sepsis rates with ultraclean airflow and body-exhaust suits, which spurred widespread adoption of laminar flow designs. Modern systems include mixed-flow, HEPA-filtered and hybrid units combining LAF with recirculation. The rationale has been to minimise bacterial load in the sterile zone by directing filtered airflow across the operative field.
The multicentre IMPACT study, evaluating 35 Italian orthopaedic ORs demonstrated effective baseline decontamination at rest, however, microbial contamination rose significantly during surgery.32 Particle counts ≥5 µm strongly correlated with microbial contamination, reaffirming their role as vectors. The study also showed that staff numbers and door openings had a nuanced effect, suggesting that human behaviour interacts in complex ways with airflow dynamics.
Evidence from Contemporary Studies
Langvantn et al(12) and Teo et al(13) separately reported that no significant differences exists between LAF and conventional ventilation in the reduction of infection risk. Conversely, Knudsen et al. reported that LAF significantly reduced airborne microbial counts compared with turbulent flow in conventional ventilation theatres during TJA.15 However, large multicentre cohorts have found no reduction in PJI with LAF in primary TJA when other infection-prevention bundles were in place, underscoring that human factors remain pivotal even with optimized ventilation.27
Particle-Based vs. Microbiological Sampling
Validation of a “clean” orthopaedic theatre has traditionally relied on bacteriological sampling, using settle plates placed near the wound and instrument tables to measure colony-forming units (CFU). In practice, many jurisdictions also reference ISO 14644-1 particle classifications and national technical memoranda (e.g., HTM 03-01) as acceptance criteria when the theatre is at rest. During surgical activity, performance is assessed by maintaining stable airflow patterns and limiting spikes in larger particle counts (≥5 µm) at the sterile field and instrument tables.33
Air quality monitoring is achieved through two complementary approaches.34,35 Optical particle counters provide real-time surveillance, reporting counts across size bins (0.3–5.0 µm) and allowing immediate corrective action when particle loads rise. Microbiological methods, such as active impaction or passive settle plates, measure viable CFU per volume or plate. While these confirm the presence of live organisms, they are retrospective, resource-intensive and prone to false positives.34,35
Consensus is shifting toward particle counting as a more practical surrogate, given its reproducibility, immediacy and strong correlation with microbial contamination for particles ≥5 µm.11,36
In orthopaedic ORs, Staphylococcus aureus and coagulase-negative staphylococci, which are common colonisers of the skin and nasal passages, are the principal airborne dispersers.37 Importantly, methicillin-resistant S. aureus (MRSA) measures approximately 0.8–1.0 µm in diameter, meaning that both larger droplets and smaller airborne particles, particularly those aggregated by static electricity, can carry high concentrations of pathogenic bacteria.36 By contrast, Gram-negative bacilli are seldom implicated in airborne transmission within this setting.20
Human Behaviour and Airborne Dispersion
Theatre staff movement within the operating theatre significantly modifies air quality and particle dispersion, making it an essential consideration in infection prevention.
Staff Movement and Particle Dispersion
Ambient particle dispersion within the OR is profoundly influenced by staff movement and the number of personnel present. Walking generates turbulence and particle release from gown hems, footwear and floor surfaces.38 Group entry into the OR produces significantly higher particle loads than staggered entry (p < 0.01), particularly in the fine particle range (0.3–1.0 µm), which is strongly associated with bacterial carriage.10,38 These turbulent flows contribute to the uplift and lateral dispersion of particles, enabling them to reach the height of instrument tables and the surgical field.
The density of staff within the OR is a major determinant of airborne contamination. Each additional person measurably increases microbial burden through the dispersal of skin squames and bacteria. Fu Shaw et al. reported an average rise of ~4.9 CFU/m³ in airborne bacteria per added individual,39 while Ljungqvist et al. estimated per-person bacterial emission rates of ~1.5–5 CFU/s depending on clothing system.40 Staff movement further amplifies this effect; Noguchi et al. demonstrated that routine activity significantly elevates airborne particle counts, and Sunagawa et al. confirmed that walking in and out of the theatre disperses particles around footwear and gown hems.18,38
Door Openings and Turbulence
Door openings represent a critical disruption to the controlled environment of the operating theatre. Each event generates transient turbulence that disperses airborne particles toward the sterile field, with measurable increases in bacterial counts even under LAF.41
Prospective studies have quantified the scale of this problem. In a Canadian series of primary TJA, mean door openings reached 71 per case over 112 minutes (0.64/min; range 0.36–1.05/min), with nursing staff responsible for 52%, anaesthesia 24% and orthopaedics 13%. Traffic peaked during the pre-incision setup phase at 0.84/min.42 Panahi et al. similarly observed that primary cases averaged 60 door openings (0.65/min), while revision cases doubled this to 135 (0.84/min),43 Similarly, Andersson et al. confirmed these patterns, showing that 64% of door openings occur pre-incision and only 36% during the procedure itself, underscoring the early phase as the key prevention target.44
Behavioural interventions can significantly reduce this burden. In a prospective two-phase study, simply posting “restricted access” signs showed a 47% reduction per case and 53% reduction per minute (p < 0.001). Even after intervention, 40 openings per case persisted over 143 minutes (0.28/min), providing a benchmark for best achievable practice.45
Surgical Helmet Systems (SHS)
SHS were introduced in the 1970s to reduce wound sepsis in TJA by providing barrier protection and directing filtered airflow away from the sterile field.46 The original Charnley whole-body exhaust-ventilated suit used a negative pressure inflow–outflow system but was limited by its cumbersome tubing, leading to the development of modern helmets. These devices draw air through a fan at the top of the helmet and circulate it within a hood and gown enclosure.47 While SHS have since become widely adopted as standard protective equipment, growing evidence suggests they may paradoxically act as sources of contamination.48,49
Baseline assessments have shown that SHS hoods can carry microorganisms even before use. Pinnoji et al. reported that 12% of hoods were contaminated at baseline, predominantly with Bacillus species, despite being supplied in “sterile” packaging.47 Contamination frequently localises around visor edges and neckline zones, areas where turbulence at helmet interfaces promotes particle accumulation. Cultures from SHS have yielded Bacillus spp., Staphylococcus epidermidis, and coagulase-negative staphylococci, underscoring their potential role as unintended reservoirs.48,50
The timing of fan activation is a critical factor influencing contamination dynamics. Computational studies have shown that activating fans before gowning disperses particles outward into the OR, whereas delayed activation after draping significantly reduces particle flux.49,50 Even under laminar airflow (LAF), glove contamination was observed in 2–3% of cases after SHS donning, highlighting that airflow systems cannot compensate for improper handling or inadequate gowning protocols.48
Rates of hood contamination rise progressively over the course of surgery, from approximately 20% at incision to as high as 80% by wound closure, depending on airflow conditions and case duration.48–50 SHS, while protective for staff, may therefore inadvertently amplify bacterial shedding into the operative field.18,19
Integrating Air Quality with Patient and Procedural Risk Factors
Air quality must also be integrated with patient- and procedure-related factors, as infection risk reflects both host vulnerability and environmental exposures.
Patient Optimization
Airborne control measures must be understood within the broader context of host vulnerability. Obese patients (BMI ≥32) show significantly higher surgical site infection (SSI) incidence due to impaired wound healing and inflammatory changes.51 While, diabetes, malnutrition and smoking similarly amplify infection risk as a result of impaired immune function.52
Importantly, these risk factors are cumulative. Dowsey et al. showed that obesity combined with diabetes markedly elevated SSI rates by 3- fold, beyond either factor alone.53 While, Kunutsor et al. further confirmed a dose-response relationship, with infection risk rising stepwise as comorbidities accumulated, reaching nearly five-fold higher in patients with ≥3 risk factors.54
These findings underscore that modifiable risk factors act synergistically, and as comorbidity burden increases, meticulous adherence to theatre air-quality measures, particularly traffic control, ventilation discipline, and strict donning protocols, becomes essential to reducing the incidence of both SSI and PJI.
Operative Time
Prolonged operative duration (>90–107 minutes) is an established risk factor for SSI, with risk rising incrementally as exposure time increases.55 Longer procedures mean greater wound exposure, increased staff movement and cumulative airborne particle generation56 Surgical efficiency therefore serves not only as a performance goal but also as a critical adjunct to environmental interventions in reducing SSI and PJI risk.54
Glove Contamination
Glove contamination parallels airborne risk, even under LAF, contamination accumulates during prolonged cases (>2 hours).41,57 Scheduled glove changes at hourly intervals, after draping, prior to cementation, and before implant handling have been shown to significantly reduce SSI risk. A systematic review by Kim et al. demonstrated that structured glove change protocols reduced contamination rates by 33–50% in prolonged TJA procedures.58 Evidence further indicates that glove perforation and contamination rates rise progressively with operative time, underscoring the rationale for routine changes.59
Staphylococcus aureus Screening
Preoperative nasal screening and decolonization with mupirocin nasal sprays, often combined with chlorhexidine washes, significantly reduces SSI and PJI rates in carriers of Staphylococcus aureus.60 Colonized patients represent a dual risk, not only are they more likely to develop endogenous infections, but they also contribute to the airborne microbial load in the operating theatre.61 Seminal investigations demonstrated that intraoperative air samples frequently isolate S. aureus strains genetically identical to those from colonized patients, confirming carriers as direct sources of airborne contamination.10,62,63 These findings underscore the importance of linking host decolonization strategies with airborne load to optimise infection prevention.
Seasonality
Arthroplasty SSI rates peak in summer months with rates in the United States increasing, up to 5.4% in August compared to 1% in winter months.64 Higher temperatures and humidity increase the rate of skin shedding and airborne microbial load, necessitating seasonal adjustments in Heating, Ventilation, and Air Conditioning (HVAC) management to mitigate risk.
Regulatory, Economic and Legal Considerations
Beyond clinical practice, the implementation of air-quality interventions is shaped by broader regulatory and economic frameworks. Compliance with national guidelines and hospital accreditation standards drives their adoption, while cost-effectiveness analyses often determine long-term sustainability. These external forces play a pivotal role in whether evidence-based measures are consistently integrated into routine orthopaedic practice.
Regulatory landscape
In 2016, the World Health Organization (WHO) issued global guidelines on SSI prevention that advised against the routine use of LAF in TJA, citing inconsistent evidence of benefit.61 This position was largely informed by registry analyses and systematic reviews showing no significant reduction in infection rates compared with conventional ventilation.65,66 The guidance sparked considerable debate within the surgical community, particularly as several national bodies continued to endorse LAF under specific conditions.
The German Society for Hospital Hygiene (DGKH), contested the WHO recommendations, arguing that the cited evidence was methodologically limited and that LAF remains appropriate for high-risk orthopaedic procedures such as TJA.67 Beyond infection outcomes, occupational health considerations also intersect with this debate as theatre air contaminants expose staff to bioaerosols, introducing medicolegal implications for hospitals and regulatory authorities. Increasingly, regulators demand transparent validation methods and robust documentation, underscoring accountability in infection prevention strategies.
Cost-Effectiveness Analysis and Return on Investment
Economic analyses demonstrate that while air quality interventions require substantial upfront investment, they offer compelling returns when weighed against PJI treatment costs. PJI treatment represents a significant economic burden, with US hospital costs projected to reach $1.85 billion annually by 2030, and individual case costs ranging from 4.5 to 5 times that of uncomplicated primary arthroplasty.68,69
LAF systems achieve cost-effectiveness thresholds when annual TJA volumes exceed 1,217 cases (assuming 10% PJI reduction) or 487 cases (assuming 25% reduction), with per-case costs decreasing from $139 in low-volume centres to $25–28 in high-volume facilities.70 Conversely, maintaining high air change rates incurs substantial costs, with each additional 5 air changes per hour costing $5,000–10,000 annually per OR. Some hospitals saved over $1 million annually by reducing air changes.71
High-efficiency particulate air and ultraviolet recirculation systems (HUAIRS) demonstrated sustained SSI reductions,60 while properly implemented LAF systems may cost as little as €3.24 per case.67 Given that PJI treatment costs range from $19,476 for debridement to >$42,000 for two-stage revision,72,73 even modest infection reduction justifies air quality investments in high-volume centres.
Future Directions
Emerging technologies show considerable promise. Ultraviolet-C (UV-C) disinfection systems reduce both surface and airborne bacterial loads, offering an additional layer of intraoperative protection.74,75 Their main limitation lies in the need for controlled exposure times and potential safety concerns for staff. Hybrid HEPA-UV recirculating units (HUAIRS) continuously filter and sterilise operating room air, providing real-time protection against airborne contamination.70 However, they require substantial upfront investment and validation in large-scale clinical studies. Automated traffic monitoring with OR cameras offer objective, real-time enforcement of traffic policies, addressing a key behavioural risk factor.76 While this improves compliance, it raises issues of cost, staff acceptance and medicolegal considerations related to surveillance.
Taken together, these innovations highlight the direction of modern air-quality management, combining engineering controls with behavioural strategies and patient optimisation. recommendations are summarised in Table 1.
Conclusion
Air quality remains a cornerstone to infection prevention in TJA. However, its benefit is maximised only when embedded within a holistic infection-prevention strategy that integrates environmental controls, staff behaviour, patient optimisation and cost-effective implementation.
While debate continues over the independent efficacy of LAF, the balance of evidence supports its selective use in high-risk procedures and high-volume centres, particularly when coupled with emerging technologies and behavioural safeguards. Ultimately, infection prevention in arthroplasty must be viewed not as a single intervention, but as a multifaceted, system-level endeavour linking environment, host, surgical technique, and institutional policy.