INTRODUCTION
Hip fracture represents one of the most serious and disabling types of traumas affecting older adults and constitutes a growing public health challenge as population aging accelerates worldwide.1 The most common hip fractures are femoral neck fractures and intertrochanteric fractures. Beyond the immediate orthopedic injury, hip fracture reflects an underlying state of biological vulnerability characterized by reduced physiological reserve, impaired musculoskeletal integrity and diminished capacity to respond to surgical stress.2 In this context, postoperative pain emerges as a central determinant of recovery, exerting a profound influence on early mobilization, participation in rehabilitation and restoration of functional independence.3 Inadequately controlled pain has been associated with delayed ambulation, prolonged hospital stay, increased risk of delirium and progression from acute to chronic postsurgical pain—outcomes that are particularly detrimental in frail elderly patients.4 Moreover, age-related changes in pain perception, polypharmacy and concerns regarding opioid-related adverse effects often result in suboptimal analgesic strategies in this population.5 Recovery after hip fracture surgery is therefore strongly shaped by the complex interplay between postoperative pain, frailty and nutritional status, which act both independently and synergistically to determine postoperative outcomes, quality of life and survival.6 Integrating pain assessment and management with geriatric and nutritional evaluation is increasingly recognized as a cornerstone of comprehensive perioperative care in older adults with hip fracture.7
Malnutrition is highly prevalent among elderly patients admitted with hip fracture and is consistently associated with adverse clinical outcomes, including increased postoperative complications, prolonged hospitalization, delayed mobilization, institutionalization and mortality.8 Age-related anorexia, chronic disease burden, inflammation and acute catabolic stress following trauma and surgery contribute to deterioration of nutritional status in this setting.9 Importantly, malnutrition frequently coexists with sarcopenia and frailty, further exacerbating functional decline and impairing rehabilitation potential.10 These interrelated conditions form a pathological continuum that compromises postoperative recovery and underscores the importance of early nutritional identification and intervention.11
In addition, postoperative delirium is a frequent and serious complication in older adults undergoing hip fracture surgery, associated with increased morbidity, prolonged hospitalization, functional decline and higher short- and long-term mortality. Its development is multifactorial, reflecting the interaction between acute surgical stress, pain, inflammation, pre-existing cognitive vulnerability, frailty and potentially modifiable factors such as inadequate pain control and poor nutritional status.12 Frailty, defined as a multidimensional syndrome of decreased physiological reserve and increased vulnerability to stressors, is highly prevalent in older hip fracture patients and serves as a robust predictor of poor outcomes, including increased complications, loss of independence and one-year mortality.13 Nutritional impairment is a core component of frailty and inadequate protein and energy intake accelerates muscle wasting, immune dysfunction and impaired wound healing.14 Within this context, pain emerges as a critical yet often underappreciated factor: Insufficient pain control may delay mobilization, limit participation in rehabilitation and contribute to persistent postoperative pain syndromes.5,15 Moreover, emerging data suggest that nutritional deficits and systemic inflammation may modulate pain perception and recovery trajectories, linking metabolic status to both acute and chronic postoperative pain.16,17 These observations align with prior work highlighting the interconnected roles of nutrition, inflammation and postoperative pain outcomes in elderly surgical patients.18
Given the high prevalence of nutritional risk in this population, validated screening tools are essential for early identification and risk stratification. The Mini Nutritional Assessment–Short Form (MNA-sf) is widely used in geriatric practice and has demonstrated strong predictive value for morbidity, functional decline and mortality in elderly surgical and hip fracture populations. Complementarily, the modified Nutrition Risk in the Critically Ill (mNUTRIC) score incorporates age, comorbidities, disease severity and inflammatory burden, offering a dynamic assessment of nutrition-related risk in acutely ill and perioperative patients. The combined use of MNA-sf and mNUTRIC may provide a comprehensive evaluation of both chronic nutritional vulnerability and acute metabolic stress, facilitating targeted nutritional and multimodal interventions aimed at improving pain control, reducing frailty progression and enhancing postoperative recovery.19
The primary objective of this noninterventional, observational, prospective study is to investigate the association between selected biomarkers and nutritional screening tools and postoperative pain outcomes—both acute and chronic—in elderly patients undergoing orthopedic surgery for hip fracture. Secondary objectives include examining the relationships between these biomarkers and nutritional parameters and frailty status and functional outcomes in patients with or without nutritional deficiencies. By addressing existing knowledge gaps, this study aims to support personalized perioperative and postoperative care, including the management of delirium and to optimize postoperative pain management and nutritional strategies, ultimately enhancing recovery, functional independence and quality of life in older surgical patients.
MATERIALS AND METHODS
This noninterventional, observational, prospective study enrolled 80 elderly orthopedic patients aged ≥80 years who underwent surgical treatment for hip fracture, including femoral neck and intertrochanteric fractures, irrespective of nutritional status. Patient recruitment occurred over a four-year period, from April 2020 to March 2024. All participants provided written informed consent after receiving comprehensive information about the study, in addition to the hospital’s mandatory consent for anesthesia and surgical intervention. Exclusion criteria comprised patients with diagnosed psychiatric disorders, active malignancy, ongoing corticosteroid or chemotherapy treatment and chronic inflammatory gastrointestinal diseases.
All patients included in the study received subarachnoid anesthesia. Prior to the induction of anesthesia, full monitoring was applied to each patient, including electrocardiogram, pulse oximetry and noninvasive arterial pressure measurement. For subarachnoid anesthesia, the same anesthetic protocol was strictly followed, receiving 12.5mg levobupivacaine and always the same type of atraumatic 25G needle (Pencan).
Preoperatively, specific laboratory tests were conducted on each patient, including those utilized for the purposes of this study: Red cell distribution (RDW), C-reactive protein (CRP), lactate dehydrogenase (LDH), calcium (Ca), ferritin (Fer) and albumin (Alb). In addition, preoperative assessments were conducted using the Visual Analog Scale (VAS), level of frailty, MNA-sf, mNUTRIC score, Sequential Organ Failure Assessment (SOFA) score and acute physiology and chronic health evaluation (APACHE) score for each patient. In the postoperative period, pain intensity was assessed using the Visual Analog Scale (VAS) to evaluate acute postoperative pain, pain at 30 days after surgery and the presence of potential chronic pain at six months. Functional independence was additionally assessed following hospital discharge.
The analytical strategy adopted in this study encompassed a comprehensive statistical evaluation designed to investigate and quantify associations between clinical predictor variables and ordinal postoperative pain outcomes. The analysis was structured into two main stages.
Ethical considerations
Ethics approval was obtained by the Committee of the Medical School of Democritus University of Thrace (DUTh) in Greece, with registration number ΔΠΘ/ΤΙΑΤΡ/35548/3207, dated 27/02/2020. The study protocol was also registered in ClinicalTrials.gov database with identification number NCT06802575. All participants provided written informed consent after receiving comprehensive information about the study, in addition to the hospital’s mandatory consent for anesthesia and surgical intervention.
Statistical Analysis
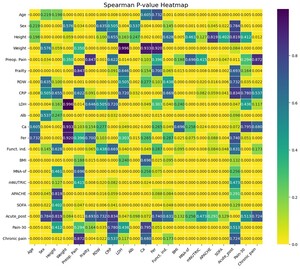
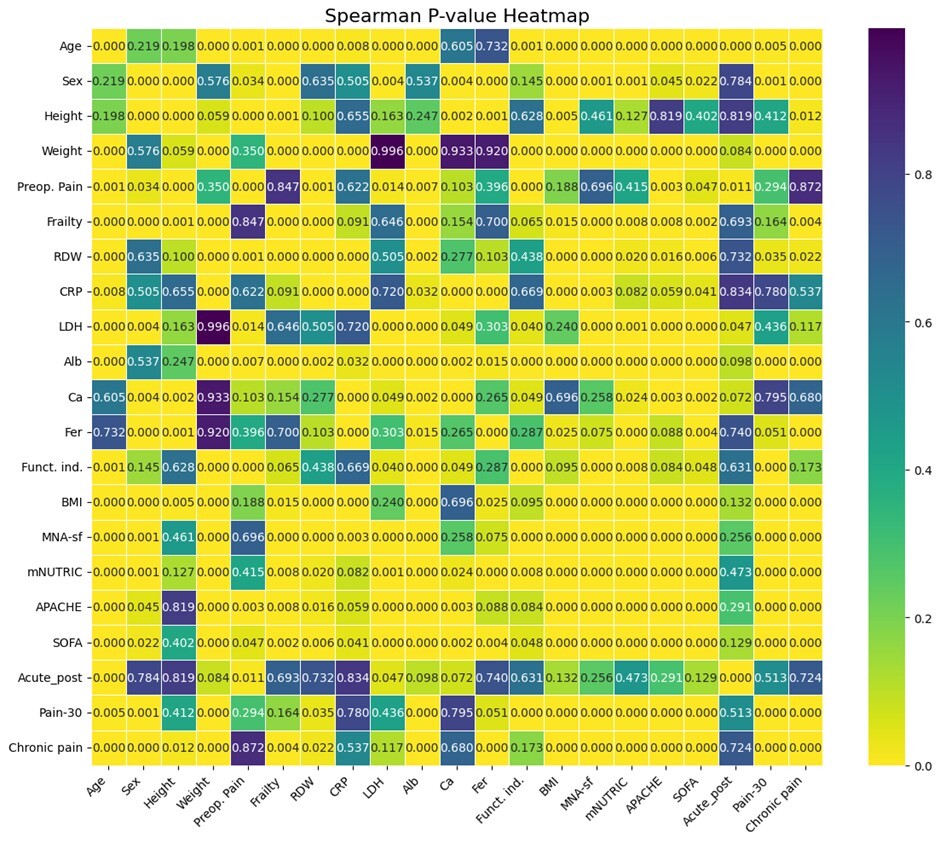
Initially, associations between all pairs of predictor and outcome variables were assessed using Spearman’s rank-order correlation coefficient, a nonparametric measure suitable for examining monotonic relationships without assuming normality or linearity of the underlying data distributions.20 Predictor variables were utilized directly as continuous or semicontinuous measures, while ordinal outcome variables were converted into numerical codes, preserving their inherent order. Statistical significance for these correlation coefficients was evaluated using corresponding p values derived from the Spearman correlation tests, applying a conventional alpha threshold of 0.05. The resulting correlation coefficients and their associated p values were visualized using heatmaps to facilitate interpretation and comparison across variables.
The results from the power analysis were systematically arranged into matrices and visualized using heatmaps, allowing clear identification of variable pairs with sufficient power to detect the observed correlations reliably. This approach facilitated an informed interpretation of the observed correlations, highlighting both robust associations and areas requiring greater statistical power, potentially through increased sample sizes or improved measurement precision in future studies.
RESULTS
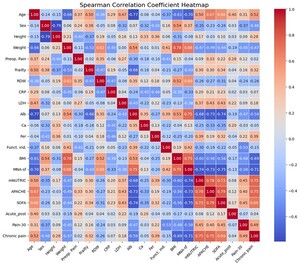
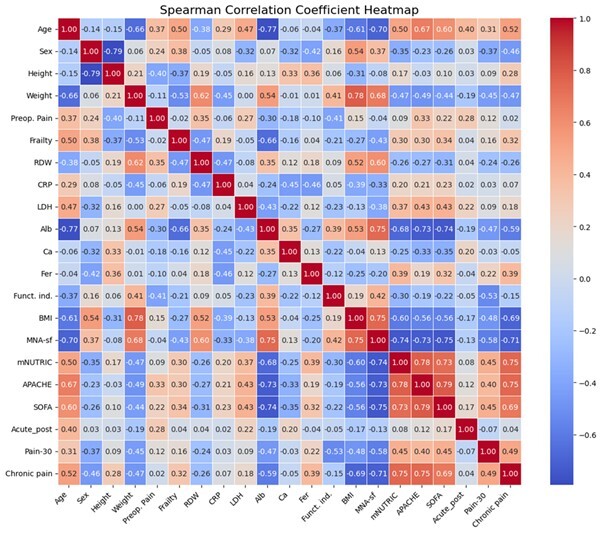
A total of 80 patients were included in the study. The analysis investigated associations among various input clinical variables (MNA-sf, mNUTRIC, APACHE, SOFA, level of frailty, RDW, CRP, LDH, Ca, Fer and Alb) as well as their relationships with target postoperative pain outcomes (presurgery pain, acute postsurgery pain, pain at 30 days postsurgery and chronic pain at six months) and functional independence after discharge. The strength of these associations was quantified using Spearman’s rank-order correlation coefficient, with statistical significance determined by p values. The results obtained using heatmaps are shown in Figures 1 and 2.
I) Association of preoperative pain with clinical, functional and biological variables
Preoperative pain was weakly associated with multiple indicators of physiological vulnerability, inflammation and disease severity. A significant weak positive correlation was observed between preoperative pain and frailty (r=0.35, p<0.001), indicating that patients with greater frailty reported higher pain levels prior to surgery. Preoperative pain also demonstrated weak positive correlations with RDW (r=0.35, p=0.001), CRP (r=0.27, p=0.014) and LDH (r=0.27, p=0.014), suggesting that systemic inflammation and metabolic stress are linked to increased preoperative pain. In contrast, Alb showed a weak negative correlation with preoperative pain (r=−0.30, p=0.007), consistent with higher pain levels among patients with poorer nutritional or existent inflammatory status. Measures of illness severity were also relevant: Preoperative pain was weakly correlated with APACHE (r=0.33, p=0.003) and SOFA scores (r=0.22, p=0.047), indicating that greater systemic illness burden is associated with higher pain prior to surgery. Very important is that validated nutritional screening tools, MNA-sf and mNUTRIC, demonstrated minimal to no statistically or clinically significant association with preoperative pain.
II) Functional Independence After Surgery
Postoperative functional independence was influenced by both pain-related and systemic variables, with effect sizes ranging from weak to moderate. Frailty demonstrated a weak negative correlation with functional independence (r=−0.21, p<0.001), indicating reduced postoperative autonomy among frailer patients. Preoperative pain showed a moderate negative correlation with functional independence (r=−0.41, p<0.001), highlighting a clinically meaningful association between baseline pain burden and poorer functional recovery. Alb exhibited a weak-to-moderate positive correlation with functional independence (r=0.39, p<0.001), underscoring the importance of nutritional and metabolic reserve in postoperative recovery. Severity-of-illness scores were also implicated: APACHE (r=−0.19, p=0.048) and SOFA (r=−0.22, p=0.048) demonstrated weak negative correlations, indicating that increased physiological derangement modestly impairs functional outcomes. Regarding the MNA-sf (r=0.42, p<0.001) and mNUTRIC (r=−0.30, p=0.008), both instruments demonstrated statistically significant correlations with preoperative pain, with a positive correlation for MNA-sf and a negative correlation for mNUTRIC, suggesting that better preoperative nutritional status contributes to better postoperative functional independence.
III) Acute postoperative pain
Acute postoperative pain showed a weak positive correlation with preoperative pain (r=0.28, p=0.011), supporting continuity of pain experience across the postoperative period. No other variables demonstrated weak or stronger associations with acute postoperative pain, suggesting that early postoperative pain is primarily influenced by baseline pain rather than systemic vulnerability or inflammatory status in this cohort. Regarding nutritional screening tools, MNA-sf and mNUTRIC demonstrated minimal to no statistically or clinically significant association with preoperative pain.
IV) Pain at 30 days post-surgery
Pain at 30 days post-surgery demonstrated stronger and more clinically relevant associations. Acute postoperative pain was moderately correlated with pain at 30 days (r=0.51, p<0.001), indicating that higher early postoperative pain is associated with persistent subacute pain. Alb showed a moderate negative correlation with pain at 30 days post-surgery (r=−0.47, p<0.001), suggesting a protective effect of better nutritional or inflammatory reserve. Systemic severity scores were also important contributors. Both APACHE (r=0.40, p<0.001) and SOFA scores (r=0.45, p<0.001) demonstrated moderate positive correlations with pain at 30 days post-surgery, linking greater perioperative illness severity to delayed pain resolution. The most important finding was that the MNA-sf (r=−0.58, p<0.001) and mNUTRIC (r=0.45, p<0.001) demonstrated moderate to strong, statistically significant correlations—negative and positive, respectively—with pain at 30 days postoperatively, suggesting that poorer preoperative nutritional status and higher nutritional risk are associated with increased postoperative pain persistence.
V) Chronic pain at six months
Chronic pain at six months showed the strongest and most consistent associations across the dataset. Pain at 30 days post-surgery was moderately correlated with chronic pain (r=0.49, p<0.001), supporting a pain chronification trajectory from the subacute to the chronic phase. Preoperative pain also demonstrated a weak positive correlation with chronic pain (r=0.32, p=0.004). Frailty showed a weak positive correlation with chronic pain (r=0.32, p=0.004), indicating that reduced physiological reserve contributes to long-term pain persistence. Alb demonstrated a moderate-to-strong negative correlation with chronic pain (r=−0.59, p<0.001), identifying low Alb as a major risk marker for chronic pain. Fer exhibited a weak-to-moderate positive correlation with chronic pain (r=0.39, p<0.001), consistent with a role of ongoing inflammatory activity. Body mass index (BMI) demonstrated a strong negative correlation with chronic pain (r=−0.69, p<0.001). The strongest associations were observed for systemic severity scores: APACHE showed a strong positive correlation (r=0.75, p<0.001) and SOFA also demonstrated a strong positive correlation (r=0.69, p<0.001), indicating that higher perioperative physiological derangement is strongly linked to chronic pain development. The most pivotal finding was that the MNA-sf (r=−0.71, p<0.001) and mNUTRIC (r=0.75, p<0.001) demonstrated strong, statistically significant correlations—negative and positive, respectively—with chronic postsurgical pain, suggesting that poorer preoperative nutritional status and higher nutritional risk are strongly associated not only with increased chronic pain severity but also with the transition from acute postoperative pain to chronic postsurgical pain.
DISCUSSION
This prospective observational study demonstrates that postoperative pain trajectories in elderly patients undergoing hip fracture surgery are strongly shaped by preoperative pain burden, nutritional status, inflammatory activity, frailty and overall physiological severity. While acute postoperative pain appears primarily driven by baseline pain intensity, the persistence of pain at 30 days and progression to chronic pain at six months are closely associated with hypoalbuminemia, systemic inflammation, frailty and illness severity. These findings extend existing models of postoperative pain by integrating nutritional and geriatric vulnerability as central determinants of pain chronification in orthopedic trauma. In other words, when negligible associations are excluded, a clear pattern emerges in which baseline vulnerability (frailty, low Alb, higher severity scores) and early postoperative pain persistence are central determinants of longer-term pain outcomes. Preoperative pain primarily influences functional recovery and later pain indirectly, whereas pain at 30 days post-surgery represents a key transitional stage with moderate predictive value for chronic pain. Nutritional status and systemic illness severity show progressively stronger associations as pain becomes persistent and chronic, supporting a time-dependent and multidimensional model of postoperative pain vulnerability in elderly orthopedic patients.
In this cohort, preoperative pain showed significant associations with frailty, inflammatory biomarkers (CRP, RDW, LDH), hypoalbuminemia and higher APACHE and SOFA scores. These findings support the concept that pain in elderly hip fracture patients reflects an underlying state of reduced physiological reserve rather than isolated nociceptive input from the fracture itself. Frailty has been consistently associated with heightened pain sensitivity and impaired stress responses in older adults. Clegg et al. and Kojima et al. demonstrated that frail individuals experience worse pain outcomes and reduced recovery following acute stressors, including surgery.2,15 In trauma patients undergoing orthopedic surgery, frailty has been linked to increased pain burden and delayed recovery after hip fracture.6,13 In hip fracture populations, frailty has consistently been identified as an independent predictor of delirium incidence, duration and severity, even after adjustment for age and comorbidities. Inadequate pain control, malnutrition and immobility—core components of the frailty phenotype—may further amplify delirium risk, highlighting the need for integrated geriatric, nutritional and analgesic strategies in this high-risk group.12,13 The observed associations between preoperative pain and CRP, RDW and LDH are consistent with evidence that systemic inflammation and metabolic stress amplify nociceptive signaling. Elevated CRP has been linked to increased pain intensity and poorer outcomes after orthopedic surgery, while RDW reflects chronic inflammatory and oxidative stress states associated with adverse geriatric outcomes.20 LDH elevation likely represents tissue injury and metabolic stress, both of which may enhance pain perception in vulnerable patients.21 The inverse relationship between Alb and preoperative pain aligns with previous studies identifying hypoalbuminemia as a marker of malnutrition and inflammatory burden associated with worse pain and functional outcomes in hip fracture patients.9,20,22 These findings support emerging evidence that nutritional impairment modulates pain perception and recovery trajectories.16,19
Postoperative functional independence was negatively associated with frailty and preoperative pain and positively associated with Alb levels. These results are consistent with prior studies demonstrating that higher pain intensity delays mobilization, limits participation in rehabilitation and predicts poorer functional recovery following hip fracture.3,4,23 Malnutrition and hypoalbuminemia are well-established predictors of impaired functional recovery, prolonged hospitalization and increased institutionalization after hip fracture.9,24,25 The findings from this study extend this literature by highlighting pain as a potential mediator linking nutritional status to functional decline. The associations between reduced functional independence and higher APACHE and SOFA scores are consistent with evidence from Chang et al. that physiological derangement and acute illness severity negatively influence postoperative recovery in elderly surgical patients.26 Acute postoperative pain showed a weak but significant association only with preoperative pain, with no meaningful correlations with nutritional or inflammatory variables. This finding aligns with existing literature identifying baseline pain intensity as one of the strongest predictors of early postoperative pain.3,17 Previous studies suggest that acute postoperative pain is primarily driven by pre-existing pain pathways and nociceptive sensitivity, whereas systemic inflammation and nutritional deficits exert greater influence on longer-term pain outcomes.27 The standardized spinal anesthesia protocol used in this cohort may also have reduced variability in immediate postoperative pain responses.
Pain at 30 days post-surgery demonstrated moderate associations with acute postoperative pain, hypoalbuminemia and illness severity (APACHE and SOFA scores). This finding is clinically important, as the subacute postoperative period has been identified as a critical window for pain resolution versus progression toward chronicity.3,17 The association between early postoperative pain and 30-day pain supports longitudinal evidence showing that inadequately controlled early pain predicts persistent postsurgical pain. The protective association of higher Alb levels is consistent with studies linking adequate nutritional and metabolic reserve to improved tissue healing and reduced inflammatory signaling.28 Higher APACHE and SOFA scores were associated with increased subacute pain, supporting evidence that greater perioperative physiological stress delays recovery and pain resolution.29
Chronic pain at six months demonstrated the strongest associations in the study, particularly with pain at 30 days, hypoalbuminemia, frailty, Fer levels, BMI and illness severity scores. These findings strongly support current models of chronic postsurgical pain emphasizing the interaction between early pain trajectories, inflammation and patient vulnerability.3,17 The strong inverse association between Alb and chronic pain highlights the central role of nutritional and inflammatory status in pain chronification. Hypoalbuminemia has been linked to chronic inflammation, sarcopenia and impaired musculoskeletal repair, all contributing to persistent pain.11,16,18 Fer, as an acute-phase reactant, has been associated with chronic inflammatory pain states, supporting its observed relationship with long-term pain.20 Frailty’s association with chronic pain is consistent with evidence that frail individuals have reduced adaptive capacity and altered pain processing, increasing susceptibility to persistent pain syndromes.2,10,15 The strong negative correlation between BMI and chronic pain likely reflects the impact of sarcopenia and undernutrition on musculoskeletal integrity and pain persistence.7,11,18 The particularly strong associations between chronic pain and APACHE and SOFA scores suggest that perioperative physiological derangement has long-lasting effects on pain outcomes. Although traditionally used to predict mortality and organ dysfunction, emerging evidence indicates that these indices also reflect vulnerability to long-term disability and pain.29
A particularly novel and clinically relevant contribution of the present study is the differential and time-dependent association of validated nutritional screening tools with postoperative pain outcomes. While neither the MNA-sf nor the mNUTRIC score showed meaningful associations with preoperative or acute postoperative pain, both instruments demonstrated progressively stronger correlations with pain persistence at 30 days and, most notably, with chronic postsurgical pain at six months. Specifically, lower MNA-sf scores and higher mNUTRIC scores were strongly associated with chronic pain development, underscoring the importance of preoperative nutritional vulnerability and acute metabolic stress in pain chronification. These findings align with emerging evidence suggesting that nutritional status does not primarily influence immediate nociceptive responses but rather modulates longer-term recovery processes, including tissue healing, inflammatory resolution and central pain processing.16–19 The inverse association between MNA-sf and chronic pain likely reflects the impact of chronic undernutrition, sarcopenia and reduced physiological reserve on musculoskeletal repair and neuromuscular function, all of which are critical for recovery after hip fracture.9–11,18 Conversely, the strong positive correlation between mNUTRIC and persistent pain highlights the role of acute illness severity and inflammation-driven catabolism in sustaining nociceptive signaling and facilitating the transition from acute to chronic pain.19,29 Previous studies have shown that mNUTRIC captures a state of heightened metabolic stress and inflammatory burden that is associated with poor outcomes in critically ill and surgical patients, but its relationship with long-term pain outcomes has been largely unexplored.19 Our findings extend this literature by demonstrating that mNUTRIC is not only a predictor of morbidity and mortality but also a robust marker of vulnerability to chronic postsurgical pain. Importantly, the opposing directions of association observed for MNA-sf and mNUTRIC emphasize their complementary roles: MNA-sf reflects baseline geriatric nutritional reserve, whereas mNUTRIC captures acute physiological stress and inflammatory risk. Their combined predictive value supports a multidimensional model in which chronic pain emerges from the interaction between pre-existing nutritional frailty and perioperative metabolic derangement. This concept is consistent with contemporary frameworks of chronic postsurgical pain that emphasize patient vulnerability, impaired recovery capacity and sustained inflammation rather than surgical factors alone.3,17 Collectively, these findings support the integration of nutritional screening tools such as MNA-sf and mNUTRIC into perioperative risk stratification, not only to predict traditional outcomes but also to identify elderly patients at high risk for pain chronification who may benefit from early nutritional optimization and targeted multimodal pain interventions.9,14,19
Limitations
Regarding limitations, although Alb is commonly used as a marker of nutritional status, it is influenced by numerous non-nutritional factors, limiting its specificity and sensitivity. Short-term changes in dietary intake may not be reflected in Alb levels, and conditions such as liver disease (e.g., hepatitis or cirrhosis), kidney disease with protein loss, and severe infection or systemic inflammation can significantly alter Alb independently of nutritional status. Consequently, Alb may better reflect overall “body reserves and stress state” rather than serve as a direct indicator of nutrition. Similarly, the APACHE and SOFA scores are not designed to assess nutritional status. While higher scores often correlate with poorer nutrition, disease severity itself may independently contribute to increased pain, potentially confounding associations between nutritional status and pain outcomes. Further limitations include the single-center design. Strengths include the prospective design, standardized anesthetic management, a relatively large cohort and longitudinal pain assessment. Nevertheless, the consistency of our findings with existing literature supports their clinical relevance.
CONCLUSION
This study demonstrates that chronic postsurgical pain in elderly hip fracture patients arises from more than surgical trauma—it reflects a convergence of frailty, nutritional vulnerability, and perioperative metabolic stress. The MNA-SF and mNUTRIC scores offer complementary insights: low MNA-SF identifies those with diminished nutritional reserves, while high mNUTRIC flags patients experiencing acute physiological and inflammatory stress. In combination with each other, they reveal a dual pathway to pain chronification, where insufficient “fuel” and excessive “fire” synergize to prolong recovery and impair function. Clinically, integrating these tools into perioperative assessment enables early, targeted interventions—combining nutritional support, multimodal analgesia, and tailored rehabilitation—to mitigate long-term pain and enhance functional independence. In essence, by addressing both the body’s reserves and its stress response, we can transform postoperative care from reactive management into proactive precision medicine, ensuring that the elderly not only survive hip fracture surgery but thrive beyond it.
Corresponding author:
Dr. Rafail Ioannidis, e-mail: raphaioan@gmail.com, phone number: 00306949418707
Authors’ Contributions
Rafail Ioannnidis: Conceived and designed the analysis, collected the data, contributed data and analysis, performed the analysis, wrote the paper
Despoina Sarridou: Conceived and designed the analysis, performed the analysis, wrote the paper, supervised the analysis
Adamantios Bampoulas: Conceived and designed the analysis, contributed data and analysis, performed the analysis, wrote the paper
Christina Tsigalou: Conceived and designed the analysis, performed the analysis, wrote the paper, supervised the analysis
Pelagia-Paraskevi Chloropoulou: Conceived and designed the analysis, performed the analysis, wrote the paper, supervised the analysis
Conflicts of Interest
There are no conflicts of interest for all the authors.