Introduction
Total hip arthroplasty (THA) is widely regarded as one of the most effective orthopedic interventions, relieving pain and restoring function in patients with advanced hip pathology. Revision THA, however, remains technically demanding, particularly in the presence of severe acetabular bone loss. Achieving durable fixation and restoring the anatomical hip center are essential to long-term success.1
Oversized acetabular components—commonly termed “jumbo” or “mega” cups—have been developed to address these challenges. By maximizing host bone–implant contact, they enhance primary stability, reduce reliance on structural grafts, and facilitate restoration of the hip center closer to its native position. This anatomical restoration improves abductor mechanics and reduces joint reaction forces.2–4
Recent advances have introduced highly porous Mega Cups, which combine the mechanical benefits of oversized shells with the biological advantages of enhanced porosity. Their interconnected architecture promotes rapid bone ingrowth and long-term osseointegration, while the increased surface area improves load distribution and reduces micromotion, thereby lowering the risk of aseptic loosening.5,6 Clinical studies and registry data increasingly support the use of jumbo and highly porous cups, reporting reliable fixation, favorable survivorship, and durable outcomes in complex acetabular reconstructions.2–7
Despite encouraging evidence, most published reports on jumbo and highly porous acetabular components are limited to single-arm case series or registry analyses. Data remains particularly scarce for patients with Paprosky type II and selected type III defects, where implant stability, graft incorporation, and long-term survivorship are most critical.8,9
The aim of the present study is to evaluate the outcomes of highly porous Mega Cups in revision THA, focusing on clinical performance, radiographic stability, and complication rates, with particular emphasis on their effectiveness in managing Paprosky type II and selected type III defects.
Patients and Methods
Study Design
This retrospective study was conducted at a single tertiary referral center between 2014 and 2022. Patients who underwent revision total hip arthroplasty (THA) with oversized, highly porous acetabular components (Mega Cup) were identified through institutional surgical records.
Ethics Approval and Consent to Participate
The study was approved by the Institutional Review Board of Mansoura University, Faculty of Medicine (Proposal Code: R.25.11.3469). All procedures were conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants for the use of their clinical and radiological data.
Patient Population
Inclusion criteria comprised patients undergoing complex or revision THA for severe acetabular bone loss requiring implantation of a Mega Cup. Exclusion criteria included incomplete medical records, follow-up shorter than 48 months, or use of alternative reconstructive techniques.
Sample size was originally calculated based on prior data indicating a mean improvement in Harris Hip Score (HHS) of 12 points (SD 18). A minimum of 18 patients was required to achieve 80% power at α = 0.05. Allowing for 10% attrition, the target sample size was set at 20 patients.
Following final screening and exclusions, 34 hips were included in the Mega Cup group. Baseline demographic and clinical variables (age, sex, BMI, indication for revision, Paprosky classification, and use of allograft or screws) were recorded.
Surgical Technique
All revision procedures were performed by experienced arthroplasty surgeons using a posterolateral approach with extensile acetabular exposure. Previous implants, necrotic tissue, and scar tissue were meticulously removed, and 3–5 intraoperative tissue samples were routinely obtained for culture.
Sequential acetabular reaming was performed until a stable press fit was achieved without compromising the acetabular columns, aiming to maximize host bone–implant contact. Host bone support was considered acceptable when ≥40–50% of the trial cup surface was engaged. The definitive acetabular component was typically 1–2 mm larger than the final reamer size.
In all cases, a cementless, highly porous oversized acetabular shell (“Mega Cup,” diameter 62–70 mm) was implanted with press fit fixation, supplemented as required with screws, augments, or morselized bone graft. Femoral reconstruction was performed using either a standard or revision stem, depending on bone quality and defect classification. Final intraoperative stability was confirmed in all cases using range-of-motion and impingement testing.
Postoperative Protocol
All patients began physiotherapy on the first postoperative day. Partial weight bearing was maintained for 6–12 weeks, with progression to full weight bearing guided by clinical and radiographic evidence of stability. Standard infection and thromboembolism prophylaxis was administered, alongside routine wound care, inpatient monitoring, and scheduled radiographic follow-up to assess osseointegration and component fixation.
Outcomes
-
Clinical evaluation: Functional outcomes were assessed using the Harris Hip Score (HHS),10 the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC),11 and the pain visual analogue scale (VAS),12 measured preoperatively and at final follow-up.
-
Radiographic assessment: Cup position, inclination, migration, and evidence of loosening were evaluated on standard anteroposterior and lateral radiographs.
-
Complications: Postoperative complications, including dislocation, infection, periprosthetic fracture, and re-revision, were systematically recorded.
-
Survivorship: Implant survival was analyzed using Kaplan–Meier survival curves, with revision for any reason defined as the endpoint.
Statistical Analysis
Continuous variables were expressed as mean ± standard deviation, and categorical variables as frequencies and percentages. Pre- and postoperative scores were compared using paired t tests. Between-variable differences (e.g., age, BMI, cup size, follow-up duration, clinical scores) were assessed using independent t tests or Mann–Whitney U tests, depending on data distribution. Categorical variables (e.g., sex, indication, Paprosky classification, allograft use, complications) were compared using chi-square or Fisher’s exact tests as appropriate. Implant survival was evaluated using Kaplan–Meier analysis with 95% confidence intervals. A p-value <0.05 was considered statistically significant. Analyses were performed using SPSS version 24.0 (IBM Corp., Armonk, NY, USA).
Results
Patient Demographics
A total of 34 hips were included in the analysis. The mean age at revision was 54.8 ± 9.2 years, and 63.6% of patients were male. The mean follow-up duration was 5.3 ± 1.8 years.
Defect severity included 72.7% Paprosky type II and 27.3% type III defects. The primary indications for revision were post infection (50.0%), aseptic loosening (31.8%), and instability (9.1%). Use of allograft and supplemental screw fixation was recorded. The mean acetabular cup size was 66.0 ± 2.5 mm, reflecting the severity of acetabular bone loss. (Table A summarizes baseline demographic and surgical characteristics.)
Implant Survival and Radiographic Outcomes
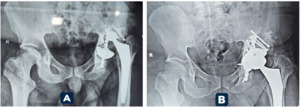
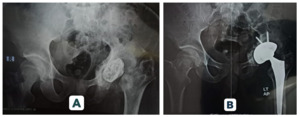
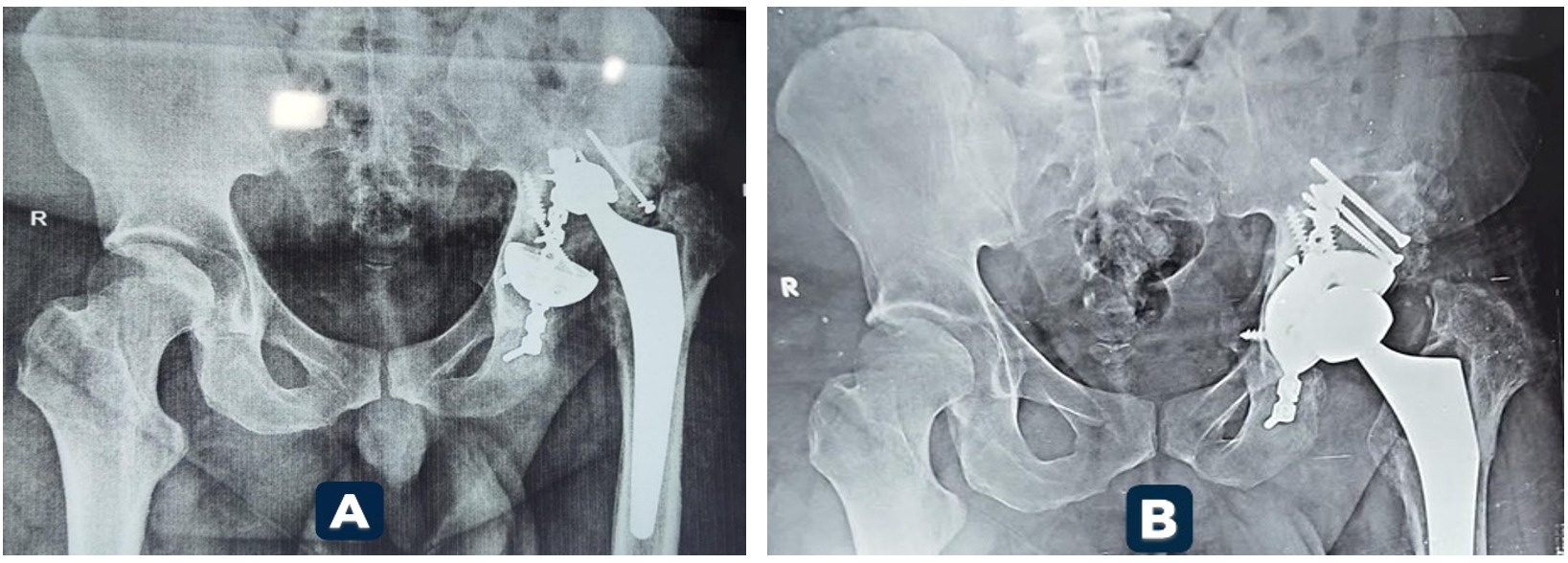
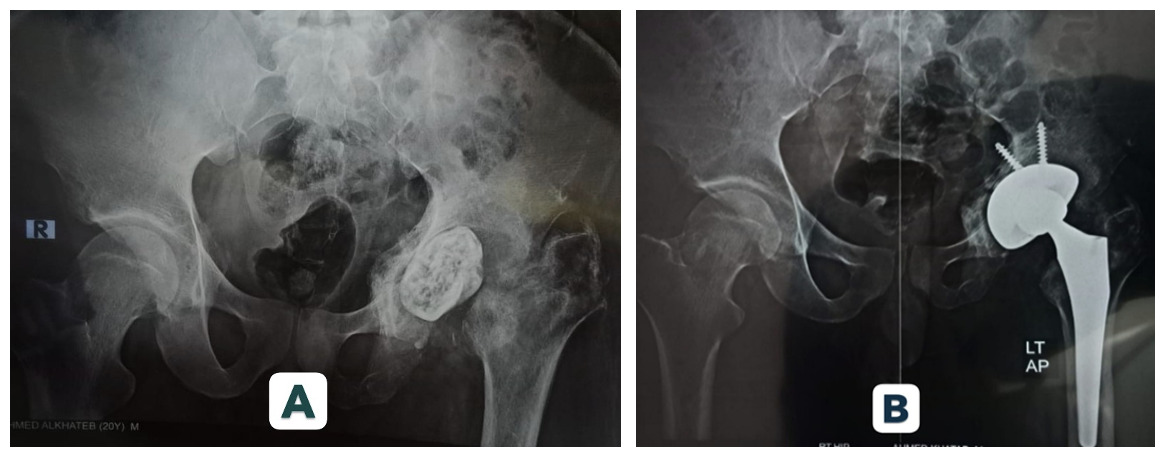
At final follow-up, 95.5% of Mega Cup reconstructions remained in situ, with no evidence of loosening. Kaplan–Meier analysis demonstrated a 95% cumulative survival rate at 5 years. Radiographic assessment confirmed stable fixation in 90.9% of hips. Graft incorporation was observed in 88.9% of cases. (Figures 1 and 2 illustrate radiographic outcomes.)
Functional Outcomes
-
Harris Hip Score (HHS): Increased from 48.2 ± 11.5 preoperatively to 82.6 ± 9.8 postoperatively (Δ +34.4, p < 0.001).
-
WOMAC: Decreased from 62.4 ± 13.2 to 24.6 ± 8.7 (Δ −37.8, p < 0.001), with consistent improvements across pain, stiffness, and function domains.
-
Pain VAS: Declined from 7.1 ± 1.4 to 2.3 ± 1.1 (Δ −4.8, p < 0.001). (Table B summarizes functional outcomes.)
Subgroup Analysis (Table C)
-
Defect Type (Paprosky):
-
Type II: 100% survivorship, 93.8% stability, HHS gain +34.4.
-
Type III: Survivorship 83.3%, stability 83.3%, HHS gain +32.7.
-
-
Allograft Use:
-
With allograft: Survivorship/stability 83.3%, HHS gain +32.3.
-
Without allograft: 100% survivorship, 93.8% stability, HHS gain +34.1.
-
Complications
-
Dislocation: 2.9%.
-
Mechanical failure: 2.9%.
-
Infection: Superficial wound infections occurred in 8.6% of patients; deep infection was observed in 2.9%.
-
Thromboembolism: 2.9%.
At final follow-up, the cumulative revision rate was 8.6%.
Discussion
Revision total hip arthroplasty (THA) in the setting of severe acetabular bone loss remains technically demanding. In this study, oversized, highly porous acetabular components (“Mega Cups”) demonstrated durable fixation, reliable survivorship, and significant functional improvement in patients with Paprosky type II and selected type III defects.
Implant Survival and Radiographic Outcomes
Kaplan–Meier analysis revealed a 95% 5-year survivorship rate, consistent with mid-term outcomes reported by Wang3 (>90%) and aligning with the favorable long-term results documented by Oommen4 at 10–14 years. Radiographic stability was achieved in 90.9% of hips, and graft incorporation was observed in 88.9% of cases. These findings parallel outcomes reported for porous titanium and tantalum constructs, reinforcing the biological advantage of highly porous shells in promoting osseointegration.5–8
Functional Outcomes
Functional recovery was substantial, with Harris Hip Score (HHS) improving by +34.4 points, WOMAC decreasing by −37.8, and pain VAS declining by −4.8. These gains exceed the typical 30–40 point HHS improvements reported in prior jumbo cup series9,13,14 and underscore the clinical relevance of highly porous Mega Cups in restoring biomechanics and patient quality of life.
Subgroup Analysis
Determinants of success highlighted the importance of residual host bone. Paprosky II defects achieved excellent outcomes, with 100% survivorship and superior functional improvement. In Paprosky III defects, outcomes were more guarded, with lower survivorship and stability, though functional gains remained clinically meaningful. The use of allograft was associated with slightly lower survivorship, consistent with variable incorporation risk, highlighting the advantage of porous shells in minimizing dependence on graft material.
Complications
Complication rates were low and within published benchmarks. Dislocation (2.9%), mechanical failure (2.9%), superficial infection (8.6%), and thromboembolism (2.9%) were observed. The overall revision rate was 8.6%, which compares favorably with rates reported for cages and more complex reconstructions.15–20
Clinical Implications
By combining mechanical stability with biological fixation, highly porous Mega Cups provide a versatile and effective option for complex revision THA. Their ability to maximize host bone contact, restore the hip center, and reduce reliance on structural grafts makes them a dependable solution in modern revision strategies.
Limitations
Limitations include the retrospective design, relatively small sample size, and mid-term follow-up. Longer prospective, multicenter studies are necessary to confirm durability beyond ten years, identify late complications, and benchmark Mega Cups against cages, triflange implants, and allografts. Registry-linked analyses could provide real-world validation and help refine patient selection criteria.
Conclusion
Highly porous oversized acetabular cups demonstrated durable fixation, reliable survivorship, and superior functional outcomes in revision THA for Paprosky II and selected III defects. The most favorable results were observed in middle-aged patients, highlighting the critical role of patient selection and defect morphology in guiding reconstruction strategies. These findings support the integration of highly porous Mega Cups into contemporary revision practice. However, in the long term, multicenter studies remain essential to establish definitive benchmarks and to validate these outcomes against alternative approaches.
Corresponding Author:
Mahmoud Mohamed Ibrahiem Abdelghafar (Mahmoud Abdelghafar), MD
Department of Orthopedic Surgery, Faculty of Medicine, Mansoura University
Algomhoria Street, Mansoura 33516, Egypt
Email: madoctor164@mans.edu.eg
Tel: +20 1008821375
Acknowledgements
The authors thank the nursing staff and physiotherapy team at Mansoura University Orthopedic Department for their support in patient care and rehabilitation
Authors’ Contributions
-
Mahmoud M. I. Abdelghafar: Study conception and design, surgical procedures, data collection, manuscript drafting, corresponding author.
-
Kamel M. A. A. Yossef: Surgical procedures, data acquisition, statistical analysis, manuscript revision.
-
Hany E. A. ElAshmawy: Data collection, radiographic analysis, manuscript editing.
-
Sallam I. Fawzy: Surgical procedures, patient follow-up, critical revision of manuscript.
All authors meet the ICMJE criteria for authorship, have read and approved the final manuscript, and agree to be accountable for all aspects of the work.
Disclosures / Conflict of Interest
The authors declare that they have no competing interests related to this study. No financial relationships, personal connections, or institutional affiliations influenced the design, conduct, or reporting of this research.
Funding
No external funding was received. The research was conducted using departmental resources without financial support from commercial or institutional sponsors.