Introduction
Degenerative disc disease is one of the leading causes of chronic lower back pain and a major contributor to the global burden of musculoskeletal disability, accounting for up to 7.7% of all years lived with disability worldwide.1 Progressive degeneration of the intervertebral discs and facet joints leads to mechanical pain, radicular symptoms, and a significant decline in patients’ functional status and quality of life.
Lumbar interbody fusion (LIF) remains a widely used and effective surgical option for degenerative, traumatic, infectious, and neoplastic spinal pathologies. Modern LIF techniques typically involve discectomy, preparation of vertebral endplates, and placement of interbody implants—cages or structural bone grafts—to restore disc height, stabilize the segment, and promote fusion.
Despite technological advancements, pseudarthrosis persists as one of the most frequent complications following LIF.2 It is defined as the absence of solid bony fusion for more than one year after surgery. Reported rates vary from 5% to 35%, with a marked increase when multilevel fusion is performed.3 Clinically, pseudarthrosis may manifest as persistent axial pain, radiculopathy, neurogenic claudication, or—less commonly—myelopathic symptoms, although in some cases it remains asymptomatic despite radiological evidence of non-union.4
Over the past decades, substantial efforts have focused on optimizing implant materials and biological enhancers to improve fusion outcomes. Nevertheless, complications such as cage subsidence and migration remain clinically relevant and continue to stimulate active research in this field.5
A wide range of interbody cage materials is currently available, including titanium, PEEK, nitinol, cobalt–chromium alloys, tantalum, and hybrid composites. Titanium and PEEK constructs remain the most extensively studied, with numerous clinical investigations demonstrating their biocompatibility and effectiveness. According to the network meta-analysis by Pairojboriboon et al., titanium cages appear to provide superior fusion rates compared with other materials.6
In parallel, various osteobiologic adjuncts—such as bone morphogenetic proteins, mesenchymal stem cells, and platelet-rich plasma—have been explored as potential enhancers of bone regeneration and interbody fusion.7
The aim of the present study was to compare the clinical and morphological effectiveness of two interbody fusion approaches using different cage materials, one of which incorporated a biocomposite hydrogel as an additional osteobiologic component.
Materials and Methods
Study Design
This prospective, parallel-group, randomized clinical study was conducted at the National Scientific Center of Traumatology and Orthopedics (Astana, Kazakhstan). The study compared two interbody fusion techniques in patients undergoing surgery for degenerative lumbar spinal stenosis. Patients were allocated in a 1:1 ratio to receive either a trabecular titanium cage combined with autologous bone and biocomposite hydrogel (Group I) or a PEEK interbody cage with autologous bone (Group II) (Figure 1).
Ethical Approval
The study protocol was approved by the Local Ethics Committee (Protocol No. 2, July 19, 2023) and conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. All participants provided written informed consent after detailed explanation of study objectives, procedures, risks, and the right to withdraw at any time without impact on their medical care. All patients were insured for the full duration of participation according to national regulatory requirements.
Eligibility Criteria
Inclusion criteria:
-
Degenerative lumbar spinal stenosis with clinical-radiological correlation
-
Segmental instability
-
Grade I–II spondylolisthesis
-
Recurrent disc herniation
-
Failure of at least 3 months of conservative treatment
-
Neurological symptoms (radiculopathy, neurogenic claudication)
-
Intervertebral disc degeneration Pfirmann grade III–V8
-
Age 30–70 years
-
Provision of written informed consent
Exclusion criteria
-
Age <30 or >70 years
-
Active infection at or near the surgical site
-
Autoimmune, psychiatric, or oncologic diseases
-
Pregnancy
-
Refusal to participate
Randomization and Group Allocation
Randomization was performed using a computer-generated sequence (simple randomization, 1:1 ratio). Allocation was managed by an independent study coordinator using sealed, opaque, sequentially numbered envelopes to prevent allocation bias.
Blinding of surgeons was not feasible due to the nature of implants; however, radiologists evaluating CT scans and fusion outcomes were blinded to group assignment.
Study Groups
Group I (n = 25): Trabecular titanium cage (Ti-6Al-4V, Grade 23) (Figure 2) filled with autologous bone graft and biocomposite hydrogel containing stromal-vascular fraction (SVF).
Group II (n = 25): PEEK cage packed with autologous bone graft (no hydrogel).
Surgical Technique
Group I — Two-Stage Protocol
Stage 1: Acquisition of adipose-derived SVF and hydrogel preparation
Adipose tissue was harvested from the anterior abdominal wall under local anesthesia using the Zuk technique.9 The lipoaspirate was transported under sterile, temperature-controlled conditions to the National Center for Biotechnology.
The biocomposite hydrogel (HCF) was prepared according to a validated protocol,10 incorporating heparin-conjugated fibrinogen, fibrinogen, aprotinin, thrombin, and calcium chloride. SVF was isolated through enzymatic and mechanical processing, washed, and expanded using standard cell culture conditions. The final cell-hydrogel composite was prepared immediately prior to implantation.
Stage 2: Posterior TLIF procedure
Under general anesthesia, TLIF was performed through a midline posterior approach. After exposure, pedicle screws were inserted at adjacent vertebrae. Decompression included facetectomy, flavectomy, and, when indicated, revision meningoradiculolysis.
The disc was removed, endplates were prepared, and the custom-designed trabecular titanium cage—preloaded with autograft and SVF-hydrogel composite—was implanted. Rods were placed and secured, and intraoperative fluoroscopy confirmed correct implant positioning. The wound was closed in layers with drainage.
Group II — Standard TLIF Procedure
Patients in the control group underwent an identical TLIF procedure, except that a standard PEEK cage filled only with autologous bone graft was placed into the interbody space.
Outcome Measures
Primary outcomes
-
Fusion rate at 12 months (CT-based Tan G.H. classification)11
-
Bone density within the cage region (Hounsfield units on CT)
Secondary outcomes
-
Pain intensity (VAS for back and leg)
-
Functional disability (Oswestry Disability Index)
-
Implant subsidence and migration
-
Intraoperative complications
-
Operative time, blood loss
Statistical Analysis
Statistical analysis was performed using SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Normality of distribution was assessed with the Shapiro–Wilk test. Continuous variables were presented as mean ± SD or median (IQR), depending on distribution.
Between-group comparisons included Mann–Whitney U test (non-parametric continuous data) and Student’s t-test (parametric data). For categorical variables Chi-square test or Fisher’s exact test were used. Repeated measures (VAS, ODI, HU dynamics) were assessed with mixed-effects model or Friedman test with Dunn–Bonferroni correction. A p-value <0.05 was considered statistically significant for all tests.
Results
Baseline Characteristics
The two study groups were statistically comparable in terms of demographic and baseline clinical characteristics (p > 0.05). Mean intraoperative blood loss did not differ significantly between Group I and Group II (292 ± 264 mL vs. 296 ± 254 mL; p > 0.05), indicating similar operative conditions (Table 1). There was not statistically significant difference in terms of sex (x2=0.08, p=0.78) or age (t(48)=1.11, p=0.27) between the two groups.
Distribution of operated levels was also similar. In Group I, the most frequent level was L5–S1 (52%), followed by L4–L5 (44%). In Group II, L4–L5 predominated (60%), followed by L5–S1 (28%). Differences were statistically insignificant (p > 0.05). No intraoperative complications were observed, and all patients were mobilized within 12 hours postoperatively.
Imaging Outcomes
Plain Radiography
Serial radiographs demonstrated correct cage positioning without migration or subsidence in all evaluated patients.
Computed Tomography and Hounsfield Unit Analysis
Osteointegration was assessed using a standardized 30-pixel circular ROI within the cage lumen and adjacent endplates. Bone density (HU) increased progressively in both groups (Table 2), indicating active fusion maturation. However, Group I consistently demonstrated higher HU values at all follow-up points.
Bone density increased significantly over time in both groups, but the titanium–hydrogel group demonstrated a markedly higher rate of HU gain, suggesting accelerated bone formation and superior osteointegration.
Local Bone Remodeling
One patient in Group II demonstrated localized endplate resorption at 6 months, without signs of instability. Partial restoration of bone structure was observed by 12 months.
Fusion Outcomes (Tan G.H. Classification)
Evaluation at 12 months demonstrated a higher proportion of complete fusion (Grade 1) in Group I compared with Group II (68% vs. 32%, p = 0.0227) (Table 3). Grades 3–4 (pseudarthrosis) were rare in both groups but occurred more frequently in controls.
These findings confirm a more favorable profile of interbody bone formation in the titanium cage + hydrogel group.
Clinical Outcomes (VAS and ODI)
Back Pain (VAS)
Both groups demonstrated significant improvement compared with baseline (Table 4), but Group I showed greater and more rapid reduction across all time points.
Leg Pain (VAS)
A similar pattern was observed for radicular pain, with significantly greater improvement in Group I (Figure 3).
Functional Disability (ODI)
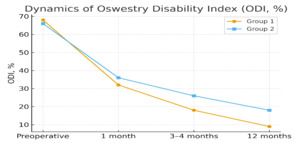
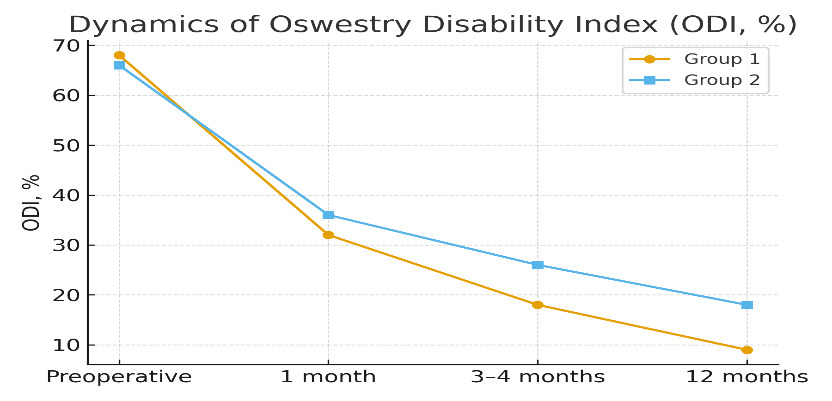
Baseline ODI scores were comparable. Group I demonstrated substantially greater improvements at 1, 3–4, and 12 months (Figure 4).
These results collectively indicate faster pain relief and functional recovery with the titanium–hydrogel technique.
Laboratory Parameters
Routine laboratory monitoring (hemoglobin, leukocytes, ESR, CRP, glucose, creatinine, liver enzymes) remained within reference ranges in both groups. Mild transient postoperative fluctuations normalized by discharge and were not clinically significant. No laboratory signs of infection or systemic inflammation were detected.
Discussion
This randomized clinical study evaluated the clinical and morphological outcomes of using a trabecular titanium cage combined with autologous bone and a stromal–vascular fraction (SVF)-based biocomposite hydrogel in comparison with a standard PEEK cage filled with autograft. The findings demonstrate that the combined titanium–hydrogel strategy resulted in more robust bone formation, higher fusion quality, and faster clinical improvement.
Superiority of the titanium–hydrogel construct in bone formation
A key finding of this study is the consistently higher Hounsfield unit (HU) values and higher rates of complete fusion (Tan grade 1) observed in the titanium–hydrogel group. The progressive increase in HU across all time points indicates accelerated mineralization and more advanced maturation of the interbody bone block. These results align with existing evidence suggesting superior osteointegration of porous and 3D-printed titanium implants compared with PEEK constructs, owing to their more favorable surface topography, wettability, and modulus of elasticity that more closely mimics trabecular bone.6,12–14
Furthermore, the biocomposite hydrogel enriched with SVF likely contributed to enhanced osteogenesis. Similar benefits were demonstrated by Choi et al.,15 who reported improved fusion parameters when SVF was used together with osteoconductive matrices. The synergy between a porous titanium scaffold and an osteoinductive hydrogel may therefore provide an optimized biological and mechanical environment for fusion.
Clinical implications
Both groups demonstrated substantial reductions in back and leg pain and improvements in disability scores by 12 months, consistent with the established effectiveness of TLIF with either titanium or PEEK cages.13,16 However, the magnitude and speed of improvement were greater in the titanium–hydrogel group. Faster early recovery may reflect more rapid stabilization of the operated motion segment or earlier onset of biological integration, which translates to reduced micromovement, diminished inflammation, and better early pain control.
While PEEK implants remain widely used due to their radiolucency and elastic modulus, their lower intrinsic osteoconductivity may partly explain the slower progression of fusion and the higher proportion of incomplete fusion grades observed in our control group.
Safety profile
No intraoperative complications were observed, and laboratory markers remained within reference ranges, supporting the safety of both surgical strategies. The transient local bone resorption detected in one control patient is consistent with known postoperative remodeling phenomena and did not affect clinical outcomes. Importantly, the addition of SVF and hydrogel did not increase infection risk or induce adverse systemic reactions, which is in line with published safety data on adipose-derived cellular products.17
Mechanistic considerations
The superior outcomes observed with the titanium–hydrogel construct may be explained by a combination of:
-
Enhanced osteoconduction due to the porous titanium architecture.
-
Improved osteoinduction from SVF-derived progenitor cells and growth factors.
-
Stabilization of the graft material through the hydrogel matrix, improving cellular retention.
-
Promotion of angiogenesis, which is critical for early integration of the interbody graft.
-
This multimodal synergy is increasingly recognized as a promising direction in interbody fusion technology.7,16
This study has several limitations.
First, it was conducted at a single center with a modest sample size (n=50), which may limit generalizability. Second, the follow-up period of 12 months, while sufficient for early fusion assessment, does not allow evaluation of long-term stability, adjacent-segment disease, or implant fatigue failure. Third, blinding of surgeons and radiologists was not feasible, introducing potential assessment bias despite the use of objective CT metrics. Fourth, the study primarily focused on radiological and pain-related outcomes; global sagittal alignment, segmental motion, and comprehensive quality-of-life instruments were not evaluated.
Finally, due to the combined use of titanium cage, autograft, and SVF hydrogel, the independent contribution of each component cannot be isolated within the present design.
Future Directions
Larger, multicenter randomized trials with long-term follow-up are warranted to confirm these findings. Comparative studies evaluating different titanium architectures, hydrogel formulations, or cellular components may help clarify optimal combinations. Additionally, incorporation of spinopelvic parameters and biomechanical modeling could provide deeper insight into how biological augmentation influences global spinal function.
Conclusion
This prospective randomized study demonstrates that the use of a trabecular titanium cage combined with autologous bone and an SVF-based biocomposite hydrogel is a safe and effective strategy for lumbar interbody fusion in patients with degenerative lumbar stenosis. Compared with a standard PEEK cage with autograft, this combined approach resulted in higher bone density within the fusion zone, a greater proportion of complete interbody fusion, and more pronounced and sustained improvements in pain and functional disability scores.
The absence of intraoperative complications and the stability of postoperative laboratory parameters support the favorable safety profile of the technique.
Overall, these findings suggest that integrating a biologically active hydrogel with a porous titanium scaffold enhances osteointegration and accelerates early clinical recovery, representing a promising direction for optimizing interbody fusion outcomes.
Acknowledgments
The authors thank the clinical and radiology teams of the National Scientific Center of Traumatology and Orthopedics named after Academician N.D. Batpenov for their support in patient care and imaging. The authors also acknowledge the engineering and laboratory teams involved in the design of the trabecular titanium cage and the preparation of the stromal vascular fraction hydrogel. This work was supported by the targeted scientific-technical program “Development and scientific justification of innovative technologies to improve the efficiency of diagnosis and treatment of injuries, consequences of injuries, and diseases of the limbs, spine, and pelvis” (BR21881815).
Author Contributions
-
Murat U. Baidarbekov – Conceptualization; Methodology; Investigation (surgery and clinical management); Resources; Supervision; Project administration; Funding acquisition; Writing – review & editing.
-
Zhangir N. Ipmagambetov – Conceptualization; Methodology; Data curation; Investigation; Formal analysis; Visualization; Writing – original draft; Writing – review & editing.
-
Nailya DeLellis – Methodology; Formal analysis (biostatistics); Validation; Supervision; Writing – review & editing.
-
Olzhas S. Bekarisov – Conceptualization; Resources; Investigation; Supervision; Project administration; Funding acquisition; Writing – review & editing.
-
Margulan S. Abdikalikov – Investigation (surgery and postoperative follow-up); Data curation; Resources; Writing – review & editing.
-
Daniyar Zh. Yestay – Investigation; Data curation; Visualization; Writing – review & editing.
Conflict of Interest
The authors declare no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
Development and scientific justification of innovative technologies to improve the efficiency of diagnosis and treatment of injuries, consequences of injuries, and diseases of the limbs, spine, and pelvis. BR21881815