Introduction
Posterolateral lumbar fusion (PLF) is preformed widely to manage degenerative lumbar spine conditions including spondylolisthesis with instability, disc degeneration with documented instability, and select cases of spinal stenosis with spondylolisthesis.1–3 As utilization of PLF continues to rise, attention has increasingly shifted from purely technical considerations to optimization of patient-specific risk factors that influence patient health and surgical outcomes.
Postoperative complications remain a concern specifically in patients with systemic risk factors such as diabetes mellitus.4 Higher rates of perioperative complications following lumbar fusion has been linked to diabetes.4–6 In PLF specifically, diabetes is associated with increased rates of adverse postoperative events, readmissions, and higher overall episode-of-care costs.7,8These complications include wound infections, delayed fusion, thromboembolic events, and cardiovascular morbidity.9,10
As surgical techniques advance, understanding risk factors that influence both short- and long-term outcomes have become increasingly important. Identifying which high-risk subgroups of patients derive disproportionate risk is essential for implementing targeted perioperative optimization strategies.
The control of diabetes in patients plays a role in determining postoperative risks.11 Poor glycemic control impairs immune function and wound healing, thereby predisposing patients to infection, nonunion, and medical complications after spinal fusion.6,12 Poorly controlled diabetes has been linked to higher rates of infection, prolonged hospitalization, and increased readmission following a lumbar spine surgery.6,11 As a result, it is important to understand the role of metabolic disease as a determinant of perioperative outcome.
Despite the recognition of diabetes as a risk factor in spine surgery, the specific contribution of diabetic neuropathy (DNP) on outcomes post PLF are unknown. Existing spine literature has largely focused on the presence of diabetes itself, without distinguishing the impact of diabetes-related complications such as neuropathy on postoperative function and complication risk. Despite the limited evidence that relates DNP with PLF, DNP is hypothesized to cause irreversible nerve damage that affects leg related outcomes.13,14 These outcomes include impaired wound healing, sensorimotor feedback, and altered pain perception.13,14
A deeper understanding of DNP and how it effects postoperative risk and recovery could help improve preoperative counseling and targeted perioperative intervention. Clarifying these relationships in the context of PLF is particularly relevant given the high prevalence of diabetes among patients undergoing lumbar fusion and the substantial resource utilization associated with postoperative complications. The purpose of this study was to evaluate the impact of diabetes mellitus and diabetic neuropathy on the complications following a single level posterolateral lumbar fusion. This was achieved through a large propensity matched cohort analysis.
Methods
Ethics
TriNetX provides de-identified data and this study was conducted without access to directly identifiable patient information, so informed consent or an IRB was not required.
Study Design and Data Source
We performed a retrospective cohort study using a multicenter database of patients undergoing single-level posterolateral lumbar fusion (PLF) between 01/01/2010 and 01/10/2026. Patients were stratified into three groups: non-diabetic, diabetic without neuropathy, and diabetic with neuropathy. Patients with incomplete demographic data or those undergoing multilevel fusion procedures were excluded.
Propensity Score Matching
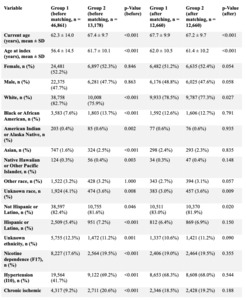
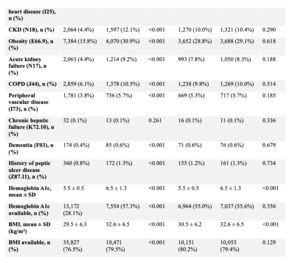
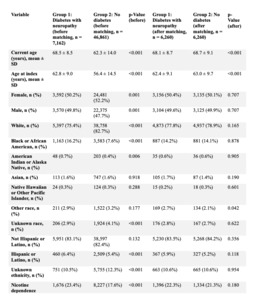
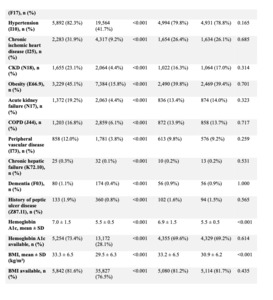
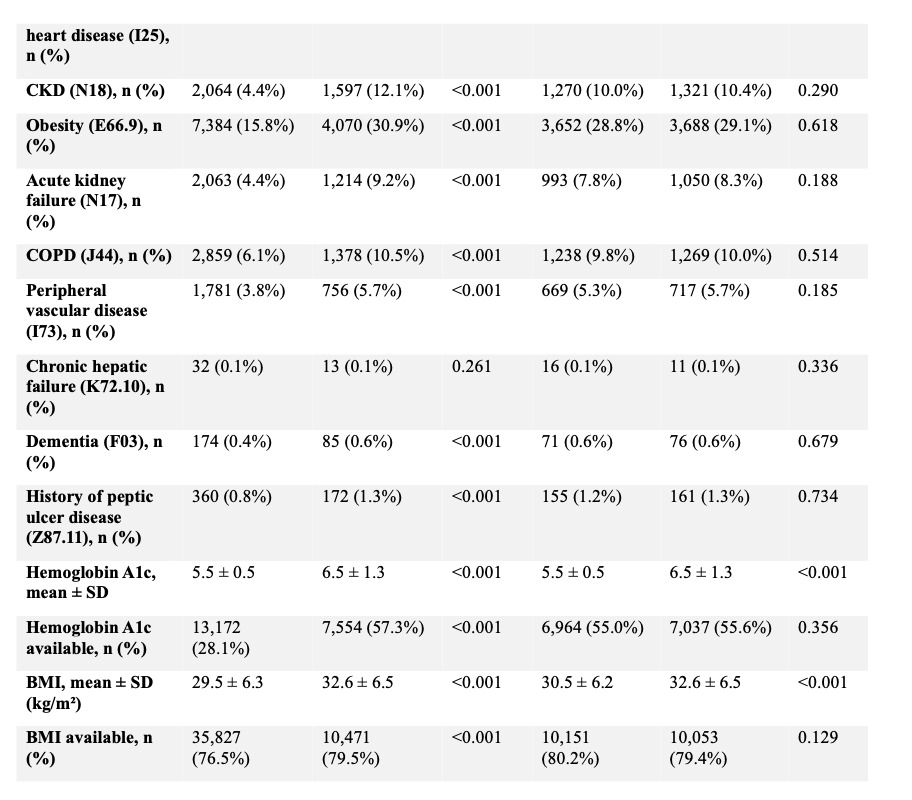
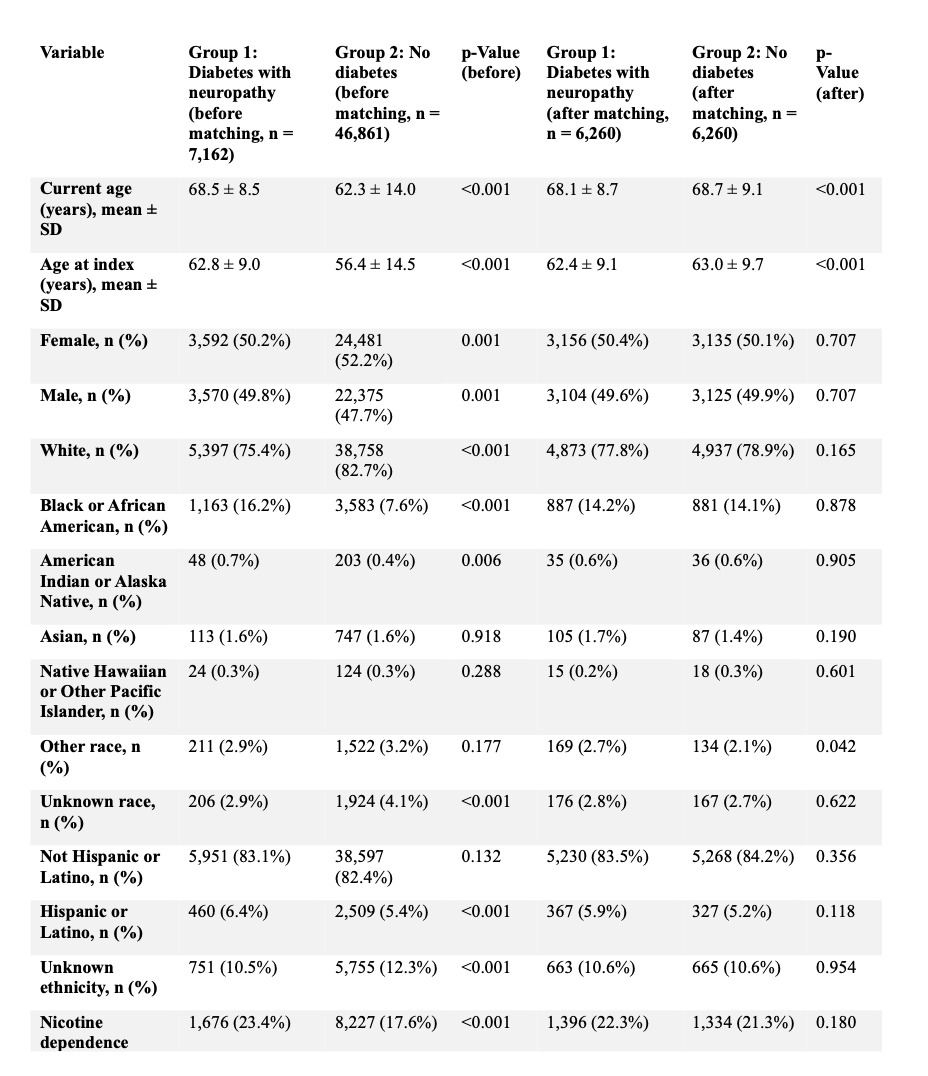
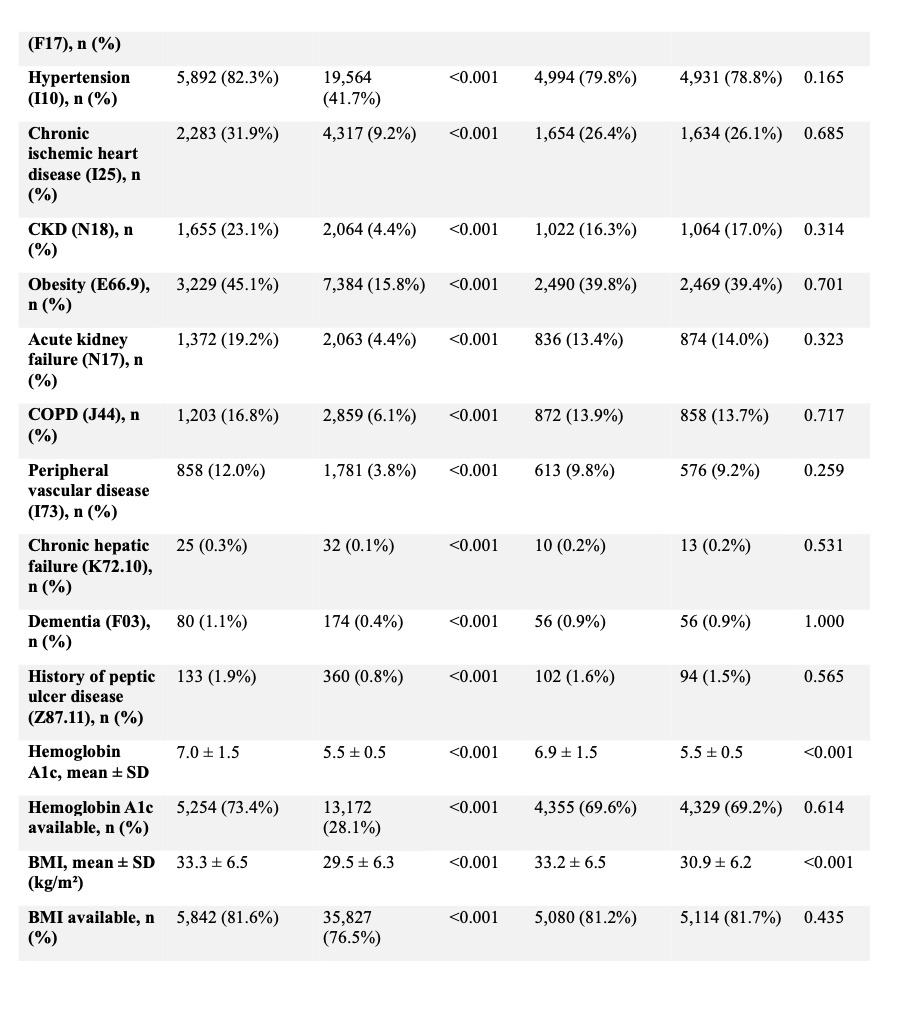
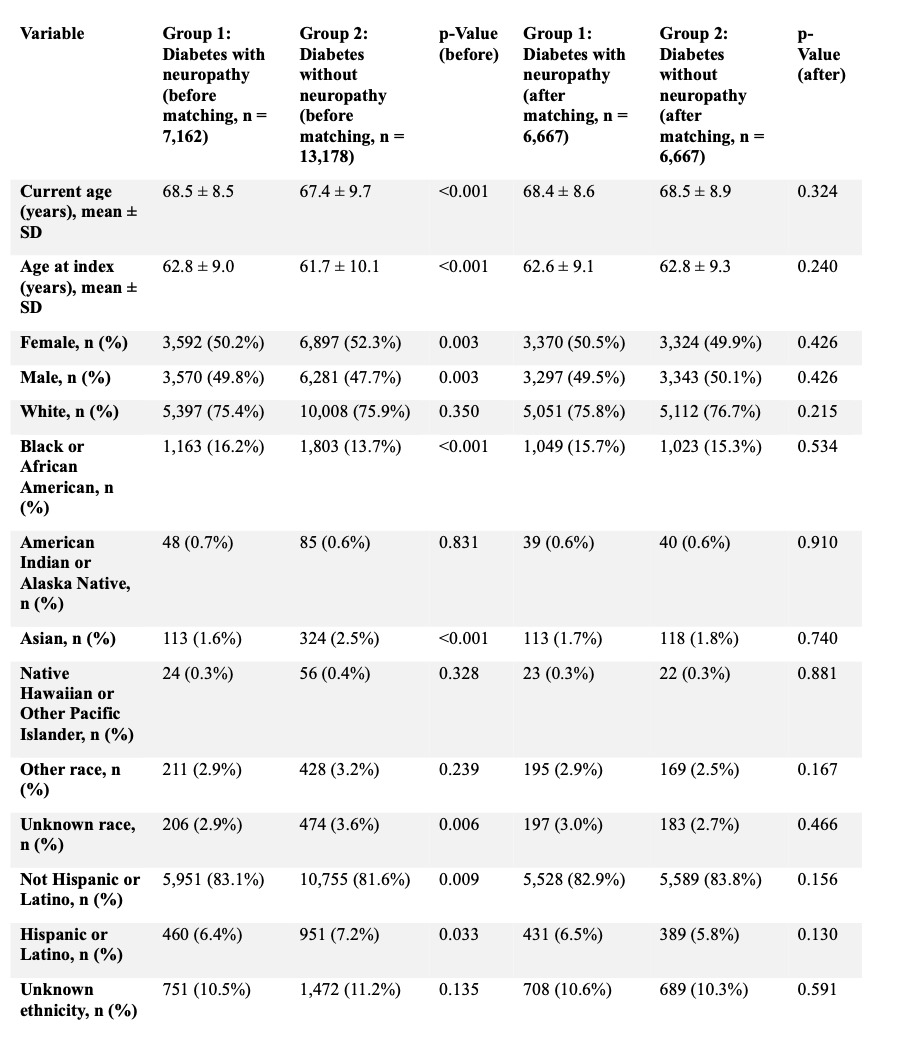
To account for baseline differences in demographics and comorbidities, propensity score matching was performed for three separate comparisons: (1) diabetic without neuropathy vs non-diabetic patients, (2) diabetic with neuropathy vs non-diabetic patients, and (3) diabetic with neuropathy vs diabetic without neuropathy. Matching was conducted using a 1:1 nearest-neighbor algorithm with a caliper width of 0.1 pooled standard deviations. Covariate balance after matching was assessed using standardized mean differences, with values <0.1 considered indicative of adequate balance; p-values are reported to describe residual differences. (Tables 1-3).
Outcomes
Primary outcomes included ninety-day postoperative medical complications (postoperative infection, deep vein thrombosis [DVT], pulmonary embolism [PE], sepsis, wound disruption, pneumonia) and two-year surgical and mechanical complications (mechanical complications, implant-associated infection, pseudarthrosis, and revision surgery). Risk ratios (RRs) and 95% confidence intervals (CIs) were calculated for all outcomes.
Results
Diabetes Without Neuropathy Versus Non-Diabetic Patients
After propensity score matching, 12,660 patients were included in each cohort with no clinically meaningful differences in baseline characteristics (Tables 1, 4, and 5).
Within ninety days postoperatively, patients with diabetes without neuropathy demonstrated higher rates of postoperative infection (5.0% vs 4.0%; RR 1.25, 95% CI 1.12–1.41; p < 0.001), sepsis (2.3% vs 1.7%; RR 1.33, 95% CI 1.12–1.58; p = 0.001), and wound disruption (3.8% vs 2.9%; RR 1.31, 95% CI 1.14–1.49; p < 0.001) compared with non-diabetic patients. Rates of DVT (1.9% vs 2.1%; RR 0.92, 95% CI 0.78–1.09; p = 0.35), PE (1.7% vs 1.5%; RR 1.10, 95% CI 0.91–1.33; p = 0.34), and pneumonia (2.0% vs 1.9%; RR 1.07, 95% CI 0.90–1.28; p = 0.44) were similar between groups. (Table 4)
At two years, diabetic patients without neuropathy demonstrated higher rates of implant-associated infection (2.9% vs 2.3%; RR 1.26, 95% CI 1.09–1.47; p = 0.003) and revision surgery (6.5% vs 5.7%; RR 1.14, 95% CI 1.04–1.26; p = 0.007), whereas rates of mechanical complications (3.0% vs 2.7%; RR 1.13, 95% CI 0.98–1.30; p = 0.10) and pseudarthrosis (15.4% vs 15.8%; RR 0.98, 95% CI 0.92–1.04; p = 0.44) were comparable. (Table 5)
Diabetes With Neuropathy Versus Non-Diabetic Patients
Following matching, 6,260 patients were included in each cohort with adequate balance across baseline covariates (Tables 2, 6, and 7). Within ninety days, patients with diabetic neuropathy experienced higher rates of postoperative infection (7.2% vs 4.6%; RR 1.55, 95% CI 1.34–1.79; p < 0.001), sepsis (3.3% vs 1.9%; RR 1.78, 95% CI 1.42–2.22; p < 0.001), wound disruption (6.0% vs 3.8%; RR 1.61, 95% CI 1.37–1.89; p < 0.001), and pneumonia (2.6% vs 2.0%; RR 1.28, 95% CI 1.02–1.61; p = 0.03). Rates of DVT (2.4% vs 2.5%; RR 0.96, 95% CI 0.77–1.20; p = 0.73) and PE (2.1% vs 1.8%; RR 1.17, 95% CI 0.91–1.50; p = 0.22) were not significantly different. (Table 6)
At two years, patients with neuropathy demonstrated higher rates of mechanical complications (3.5% vs 2.8%; RR 1.25, 95% CI 1.03–1.53; p = 0.02), implant-associated infection (4.6% vs 2.6%; RR 1.78, 95% CI 1.47–2.16; p < 0.001), and revision surgery (8.1% vs 6.2%; RR 1.31, 95% CI 1.15–1.49; p < 0.001). Pseudarthrosis rates were similar (16.6% vs 16.4%; RR 1.01, 95% CI 0.94–1.10; p = 0.75). (Table 7)
Diabetes With Neuropathy Versus Diabetes Without Neuropathy
Direct comparison of 6,667 matched pairs showed no significant residual imbalance in baseline characteristics (Tables 3, 8, and 9).
Within ninety days, diabetic neuropathy was associated with increased rates of postoperative infection (7.3% vs 5.5%; RR 1.33, 95% CI 1.17–1.52; p < 0.001), wound disruption (6.1% vs 4.3%; RR 1.41, 95% CI 1.22–1.63; p < 0.001), and implant-associated infection (4.6% vs 3.3%; RR 1.38, 95% CI 1.16–1.63; p < 0.001). Rates of DVT (2.5% vs 2.2%; RR 1.13, 95% CI 0.91–1.40; p = 0.28), PE (2.1% vs 1.9%; RR 1.11, 95% CI 0.88–1.41; p = 0.39), sepsis (3.3% vs 2.9%; RR 1.13, 95% CI 0.93–1.36; p = 0.21), and pneumonia (2.7% vs 2.5%; RR 1.06, 95% CI 0.87–1.31; p = 0.55) were similar. (Table 8)
At two years, patients with diabetic neuropathy demonstrated higher rates of revision surgery (8.2% vs 6.7%; RR 1.23, 95% CI 1.09–1.38; p = 0.001) and implant-associated infection (4.6% vs 3.3%; RR 1.38, 95% CI 1.16–1.63; p < 0.001), while rates of mechanical complications (3.5% vs 3.2%; RR 1.10, 95% CI 0.92–1.32; p = 0.31) and pseudarthrosis (16.7% vs 16.5%; RR 1.02, 95% CI 0.94–1.10; p = 0.69) were comparable. (Table 9)
Discussion
The purpose of this study was to evaluate the impact of diabetes mellitus and diabetic neuropathy on short and long term outcomes following single level posterolateral lumbar fusion. While diabetes is a well established risk factor for postoperative complications in spine surgery, the specific contribution of diabetic neuropathy has not been well defined in patients undergoing PLF. By stratifying patients into non diabetic, diabetic without neuropathy, and diabetic with neuropathy cohorts using propensity matched comparisons, this study sought to clarify whether neuropathy identifies a subgroup of diabetic patients with disproportionate perioperative and longer term risk.
In this analysis, we found that diabetes without neuropathy was associated with modest but measurable increases in postoperative complications compared with non diabetic patients. Within ninety days, patients with diabetes without neuropathy had higher rates of postoperative infection (5.0% vs 4.0%), sepsis (2.3% vs 1.7%), and wound disruption (3.8% vs 2.9%), while rates of thromboembolic events and pneumonia were similar. At two years, this group demonstrated higher rates of implant associated infection (2.9% vs 2.3%) and revision surgery (6.5% vs 5.7%), with similar rates of mechanical complications (3.0% vs 2.7%) and pseudarthrosis (15.4% vs 15.8%).
Patients with diabetic neuropathy demonstrated a more pronounced increase in both early and late complications. Within ninety days, this group experienced higher rates of postoperative infection (7.2% vs 4.6%), sepsis (3.3% vs 1.9%), wound disruption (6.0% vs 3.8%), and pneumonia (2.6% vs 2.0%) compared with non diabetic patients. Over two years, diabetic neuropathy was associated with increased rates of mechanical complications (3.5% vs 2.8%), implant associated infection (4.6% vs 2.6%), and revision surgery (8.1% vs 6.2%), while pseudarthrosis rates were similar (16.6% vs 16.4%).
When diabetic patients with and without neuropathy were compared directly, neuropathy remained associated with higher rates of early postoperative infection (7.3% vs 5.5%), wound disruption (6.1% vs 4.3%), and implant associated infection (4.6% vs 3.3%). At two years, patients with neuropathy demonstrated higher rates of revision surgery (8.2% vs 6.7%) and implant associated infection (4.6% vs 3.3%), while mechanical complications and pseudarthrosis were comparable between diabetic cohorts.
These findings suggest that diabetic neuropathy represents a marker of more advanced or systemic disease rather than diabetes alone. Prior spine literature has often treated diabetes as a binary exposure, without distinguishing the presence of diabetes related complications such as neuropathy.15,16 Our results suggest that this distinction is clinically meaningful, as neuropathy appears to capture a subset of patients with greater susceptibility to infectious and implant related complications following PLF.17,18
From a clinical standpoint, these findings have important implications for risk stratification and perioperative planning. Identification of diabetic neuropathy during preoperative evaluation may provide additional prognostic information beyond the diagnosis of diabetes alone and should be considered during surgical counseling.19 The elevated short term complication rates observed in patients with neuropathy support closer postoperative surveillance and a lower threshold for early intervention.11,20 The increased long term rates of implant associated infection and revision surgery in this group may also prompt consideration of construct selection, bone quality, and extended follow up. For patients with diabetes without neuropathy, the more modest increase in risk supports a nuanced discussion that acknowledges diabetes related risk without overstating its impact.
Several limitations of this study should be acknowledged. The retrospective design and reliance on administrative data introduce the possibility of diagnostic and coding inaccuracies and limit the ability to account for important clinical variables. Factors such as glycemic control, duration and severity of diabetes and neuropathy, and specific surgical or technical details were not available and may influence both complication risk and interpretation of these findings. Neuropathy was identified based on diagnostic coding and may not fully reflect disease severity. Future prospective studies incorporating detailed metabolic and neurologic assessments, standardized definitions of neuropathy, and more granular surgical data are needed to validate these findings and to better define targeted perioperative optimization strategies for patients with diabetes and diabetic neuropathy undergoing posterolateral lumbar fusion.
Conclusions
In this analysis, patients’ with diabetes mellitus undergoing PLF were associated with a modest increase in postoperative risk compared to non diabetic patients. This increase in risk included both an increase in short and long term complications. When comparing diabetic patients to patients within a subgroup however, those who also had diabetic neuropathy experienced higher risks of infection, wound complications, and revision surgery. There were similar rates of pseudarthrosis amongst all the cohorts, which suggests that the increase in revision surgeries within the neuropathy subgroup was not attributed to pseudarthrosis.
These findings support the idea of adopting diabetic neuropathy as a meaningful clinical marker. Preoperative risk stratification, surgical counseling, and postoperative management should take into account a diabetic neuropathy diagnosis and not just the binary presence or absence of diabetes. Further investigation is warranted to validate these findings and help mitigate the risk in this high risk population.
Authors’ contributions
All authors meet ICMJE authorship criteria and approve the final manuscript.
Abdel Kareem Hilo: Conceptualization; Study design; Data acquisition/curation; Statistical analysis; Interpretation of results; Drafting the manuscript.
Yahya Haj Hamad: Study design; Interpretation of results; Critical revision of the manuscript for important intellectual content.
Tyler Forbes: Study design; Interpretation of results; Critical revision of the manuscript for important intellectual content.
Alqasim Elnaggar: Data acquisition/curation; Methodology; Interpretation of results; Critical revision of the manuscript.
Hamza Mohammad: Interpretation of results; Critical revision of the manuscript for important intellectual content.
Bashar Jawich: Study design; Supervision; Interpretation of results; Critical revision of the manuscript.
Tarek Harhash: Had access to the study data and took responsibility for the integrity of the data and accuracy of the analysis; Project administration; Manuscript guarantor.
Ahmad Almaat: Critical revision of the manuscript.
Haytham Alqasmi: Critical revision of the manuscript.
Yuyang Chu: Project Administration, Interpretation of results; Critical revision of the manuscript for important intellectual content.
Rahul Vaidya: Project Administration, Interpretation of results; Critical revision of the manuscript for important intellectual content.
Disclosures / conflicts of interest
Each author completed conflict-of-interest disclosure consistent with ICMJE recommendations.
Conflicts of interest
The authors declare no conflicts of interest related to this work.
Funding / support
This work received no specific external funding.
Ethics
This study used de-identified data; informed consent and IRB review were not required, per the data platform’s de-identification model.


_vs._plf_w.png)
_vs._plf_w.png)






_vs._plf_w.png)
_vs._plf_w.png)




