Background
Total knee arthroplasty (TKA) is a common and effective surgery that is used to treat conditions such as advanced knee osteoarthritis and long-standing knee pain.1 It helps to improve movement, reduce pain, and help the patients to return to their daily activities. In the recent years the number of people who need knee replacement increase continuously (upto 17%), and TKA has become one of the most frequently performed orthopedic procedures worldwide.2 Although this surgery has a high success rate, but complications of this surgery can still occur. One of the important complications of this procedure is nerve injury, although it is not common (0.12%).3
The nerve that injured after the TKA can lead to several problems which affect both the recovery and long-term functioning of the muscles. These injuries can range from the mild numbness to more serious problems which included the weakness of muscles or the inability to lift the foot which is known as drop foot.4 The most commonly affected nerves was common peroneal nerve and the saphenous nerve, while the least commonly affected nerve was the tibial nerve.5 Due to the injury of these nerves, the patients may experience symptoms such as tingling, loss of sensation, burning pain, difficulty in walking, or reduction in the control of foot and ankle movements. While some patients recover fully over the time, there are some patients who may continue to have symptoms that limit mobility and daily functioning.
There are several factors which can contribute to nerve injury during or after the TKA. These include the stretching or pressure on the nerve during the surgery, the use of the tourniquet, bleeding around the nerve, correction of the severe knee deformities, and already existing nerve disease.6 The common peroneal nerve is especially at risk because of its location around the fibular head, where it can stretch easily or compressed. The saphenous nerve, which runs along the incision line, can also be affected during normal surgical exposure.7 Despite the known causes, the exact rate of nerve injury after TKA varies widely across published studies. There are some reports which show very low rates (0.12%),3 while others show higher numbers of injuries (4.3%), depending on how nerve injury is defined and how long patients are followed after surgery.7
In recent years, there have been improvements in the surgical technique, anesthesia, and the postoperative care that aim to reduce the occurrence of complications after TKA.8 However, the evidence on whether nerve injury rates have decreased remains unclear. There are different studies which reported variable findings, and there is no single, consistent evidence of how often nerve injuries occur or which factors increase the risk of these injuries. These inconsistencies make it very difficult for the surgeons to provide an accurate information to patients and to plan preventive strategies.
There is a need for the systematic review and meta-analysis in order to bring all available evidence together in a single study. This systematic review and meta-analysis aim to summarize the current evidence on the nerve injuries after the total knee arthroplasty. The goal is to provide the clear and updated picture of how often these injuries can occur, what types of nerve injuries are most common after the TKA, and what factors may increase the risk of these injuries. This information can support the surgeons and healthcare teams in the improvement of the patient safety and optimizing outcomes after TKA.
Materials and Methods
This study was designed as a systematic review and meta-analysis, and was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guideline
Eligibility Criteria
Studies were included if they met predefined eligibility criteria. The population of interest comprised adult patients (≥18 years) who had undergone primary or revision total knee arthroplasty (TKA) for any indication. Eligible studies examined TKA procedures, including variations in implant type or anesthesia method, and reported postoperative nerve injuries—such as common peroneal nerve palsy, femoral nerve injury, saphenous nerve injury, tibial nerve injury, or other peripheral neuropathies—occurring as direct complications of TKA. We included randomized controlled trials, cohort studies, case-control studies, and large case series with at least 10 patients. Only articles published in English were considered. Studies were excluded if they were case reports or case series involving fewer than 10 patients, cadaveric, animal, or biomechanical investigations, conference abstracts without available full text, or studies that did not specify nerve injury outcomes or were unrelated to TKA.
Search Strategy
The literature search was conducted primarily using PubMed and Google Scholar. Additionally, we performed manual reference checks of relevant reviews and included studies to ensure comprehensive identification of eligible publications. The search strategy incorporated both Medical Subject Headings (MeSH) and free-text terms related to TKA and nerve injury.
Study Selection
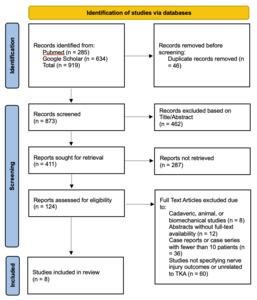
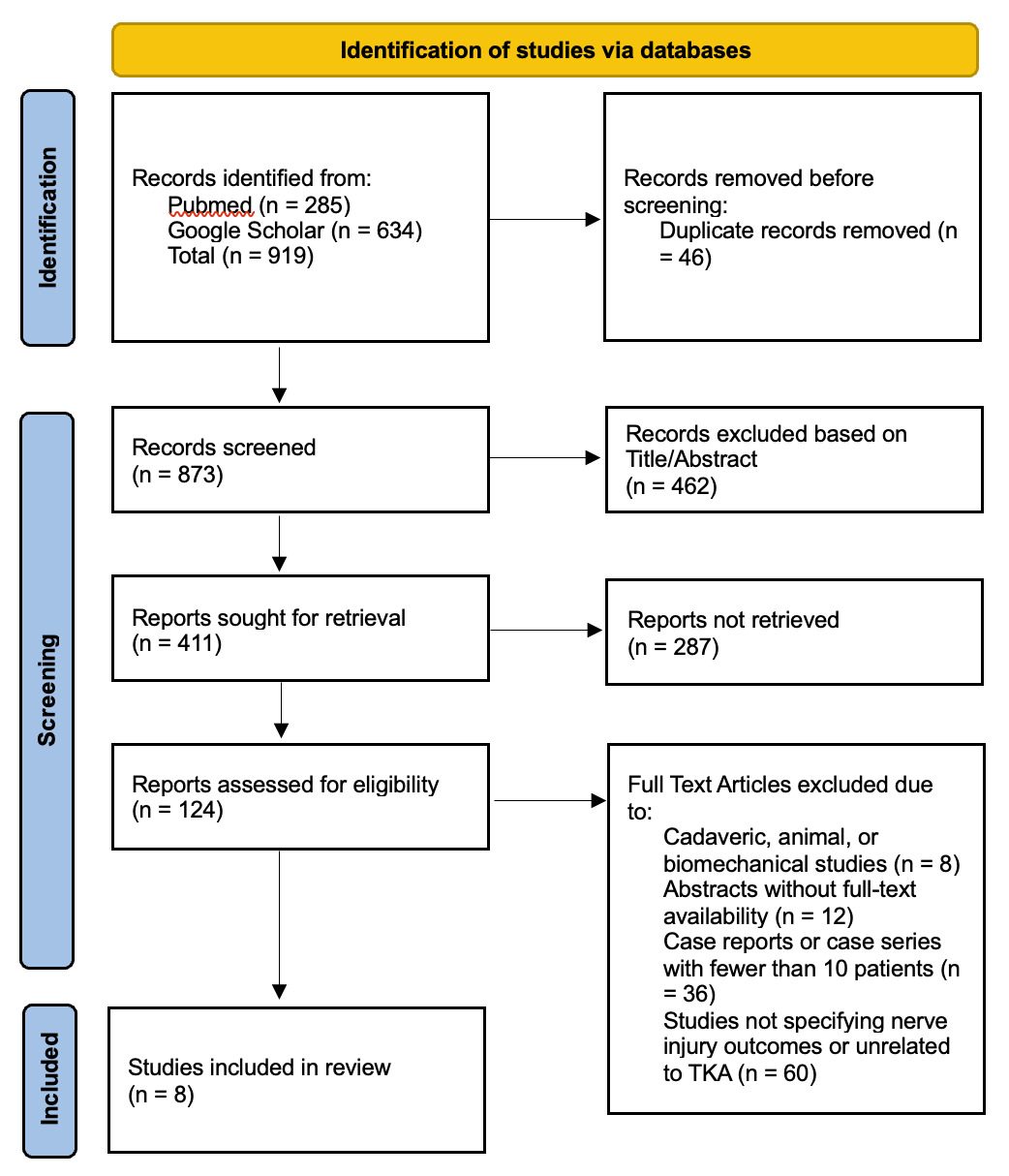
All identified records were imported into EndNote (Clarivate Analytics) for duplicate removal. Two independent reviewers screened the titles and abstracts to identify potentially eligible studies, after which the full texts of selected articles were retrieved and assessed according to the predefined inclusion and exclusion criteria. Any disagreements were resolved through discussion or, when necessary, consultation with a third reviewer. The study selection process is summarized in the PRISMA flow diagram (Figure 1), which illustrates the number of records identified, screened, excluded (with reasons), and the final set of studies included in this review.
Data Extraction
Data extraction was carried out independently by two reviewers using a standardized Microsoft Excel form. Any discrepancies between the reviewers were addressed through discussion until consensus was reached.
Risk of Bias and Quality Assessment
The methodological quality of the included studies was thoroughly assessed with the help of two validated tools, the Newcastle-Ottawa Scale (NOS) for observational studies. This evaluation was conducted independently by three reviewers. Any disagreements were resolved through consensus, and if needed, a senior reviewer was consulted to ensure objectivity. The summarized results are shown in Figures 2 & 3. Notably, all the included studies demonstrated acceptable methodological quality. Most studies showed low risk in cohort representativeness and exposure ascertainment, consistent with established orthopedic cohort assessments such as those by Schinsky et al. and Park et al. Outcome assessment was robust across studies, reflecting methods comparable to prior large-scale TKA investigations such as those by Vince and Chmell, who emphasized consistent neurological examination and EMG confirmation in postoperative neuropathy research. However, the comparability domain showed moderate concerns, as several studies lacked adjustment for key confounders, echoing limitations described in earlier observational analyses of TKA complications (e.g., Idusuyi et al. and Speelziek et al.). A few studies also showed unclear follow-up adequacy, a challenge frequently noted in long-term arthroplasty outcome literature, such as the work of Dy et al. on postoperative nerve deficits. Despite these limitations, the overall quality was sufficient to support confidence in the pooled meta-analytic findings.
Results
Our review included eight studies for the assessment of prevalence of nerve injuries after total knee arthroplasty. Notably, the majority of studies originated from the United States, with limited representation from other regions which included multicenter study from the USA, Netherlands, and Taiwan by Varsseveld (2025)9 and a Norwegian cohort by Hovik (2024).10 The retrospective cohort designs were most common,9–12 while Jayaram (2025)13 contributed a large retrospective database study and Shetty (2018)14 provided a case–control design. The period of studies ranged from recent years (2010–2024)9,13 to long-term datasets such as Speelziek (2019)11 and Idusuyi (1996).15 Follow-up duration ranged from acute assessment14 to more than three years.11,15 Sample sizes varied widely, from 26 patients in Varsseveld (2025)9 to over 1.5 million cases in Jayaram (2025)13 (Table 1).
Notably, the age of the patients was generally in the mid-60s, with median or mean ages ranging from 61.5 to 69.6 years.9–16 Most cohorts showed that there is a female predominance such as Jayaram (2025)13 and Shetty (2018).14 BMI values were consistently in the overweight to obese range, with reported means or medians between 28 and 35 kg/m².9,10,12,14 The common comorbidities included diabetes mellitus, reported in 4%–21% of patients across several studies,9–12,14–16 along with psychological conditions, autoimmune disease, hypothyroidism, and spinal disorders depending on the cohort. The preoperative deformity patterns showed that valgus deformity commonly represented in studies such as Speelziek (2019)11 and Park JH (2013),12 while others studies reported mixed varus–valgus distributions.9,10,15,16 Most procedures were primary TKAs, with the revision rates generally below 10%.9,12–15 Laterality reporting was inconsistent, though available data showed both unilateral and bilateral involvement9,15,16 (Table 2).
Regarding the surgical and perioperative practices, the commonly reported approach was medial parapatellar, which is reported by Hovik (2024)10 and Park JH (2013),12 while Varsseveld (2025)9 also described lateral incisions for the cases which involved the common peroneal nerve. The type of implant ranged from cruciate-retaining and posterior-stabilized designs in Hovik (2024)10 and Schinsky (2001)16 to constrained components in the older cohorts such as Idusuyi (1996).15 Tourniquet use was frequent, with durations typically between 58 and 129 minutes.10,12,14–16 Anesthesia modalities varied, with combinations of the epidural, spinal, and general anesthesia reported across studies.10–12,15,16 Moreover, peripheral nerve blocks were rarely reported by Varsseveld (2025)9 and Speelziek (2019).11 Preoperative deformity correction ranged from mean angles of 9° to over 12° in several cohorts.9,10,12,15,16 Prophylactic nerve release was rare, documented only in isolated cases.11 Surgeon experience ranged from high-volume specialized centers to multi-surgeon cohorts9,14–16 (Table 3).
Notably, the most frequently affected nerve was the common peroneal nerve, which is reported consistently in multiple cohorts,10–12,14–16 while some studies also identified saphenous or IPBSN involvement9 and occasional sciatic or tibial injuries.11,14 The reported incidence varied from 0.16% to 1.3% depending on the sample size and diagnostic criteria.10–16 Most injuries were typically unilateral and some cases were bilateral.9,15,16 Onset commonly occurred immediately or within the first few postoperative days,10–12,14–16 although delayed presentations were documented, such as a median onset of 29.5 months was reported by Varsseveld (2025).9 The clinical presentations frequently included foot drop, sensory deficits, neuropathic pain, or combined motor–sensory loss.9–12,14–16 These injuries were diagnosed based on clinical examination supplemented by EMG, imaging, or coding data in large cohorts.9–16 The severity of injury ranged from incomplete palsy to complete deficits, with several studies reporting a mix of both.10,12,15,16 Intraoperative findings were uncommon but included hematomas and valgus-correction–related tension in selected cases11,15 (Table 4).
The initial management of these nerve injuries involved conservative measures such as loosening dressings and positioning the knee in 20–30° flexion.10,12,15,16 Some studies only used observation alone without surgery.10,14,16 Moreover, the surgical intervention was selective, which included neurectomy or the decompression in Varsseveld (2025),9 nerve releases and hematoma evacuation in Speelziek (2019),11 and the immediate evacuation for compressive lesions in Park JH (2013).12 Timing of the management ranged from early postoperative intervention to delayed surgery months after TKA.9,11,15 Adjuvant therapies were rarely reported, aside from isolated steroid or neuropathic pain treatments.11,12 Rehabilitation commonly included AFO support and physiotherapy10,12,15,16 (Table 5).
The recovery outcomes of these nerve injuries showed substantial improvement. Varsseveld (2025)9 showed 95.5% recovery with improved pain and the functional scores. Hovik (2024)10 reported 68% full recovery within 6–24 months, with few persistent deficits. Speelziek (2019)11 observed 78% full or near recovery in about 10 months, while Park JH (2013)12 and Schinsky (2001)16 reported similar recovery rates of 67–68%, with around one-third retaining partial deficits. Idusuyi (1996)15 found that equal numbers of full and incomplete recoveries, with long-term deficits in several cases. Orthotic use was common in persistent weakness, and reoperations were rare (Table 6).
Meta-Analysis of Assessment of Nerve Injuries After TKA
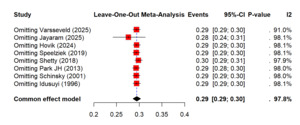
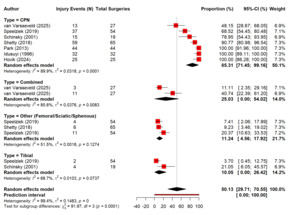
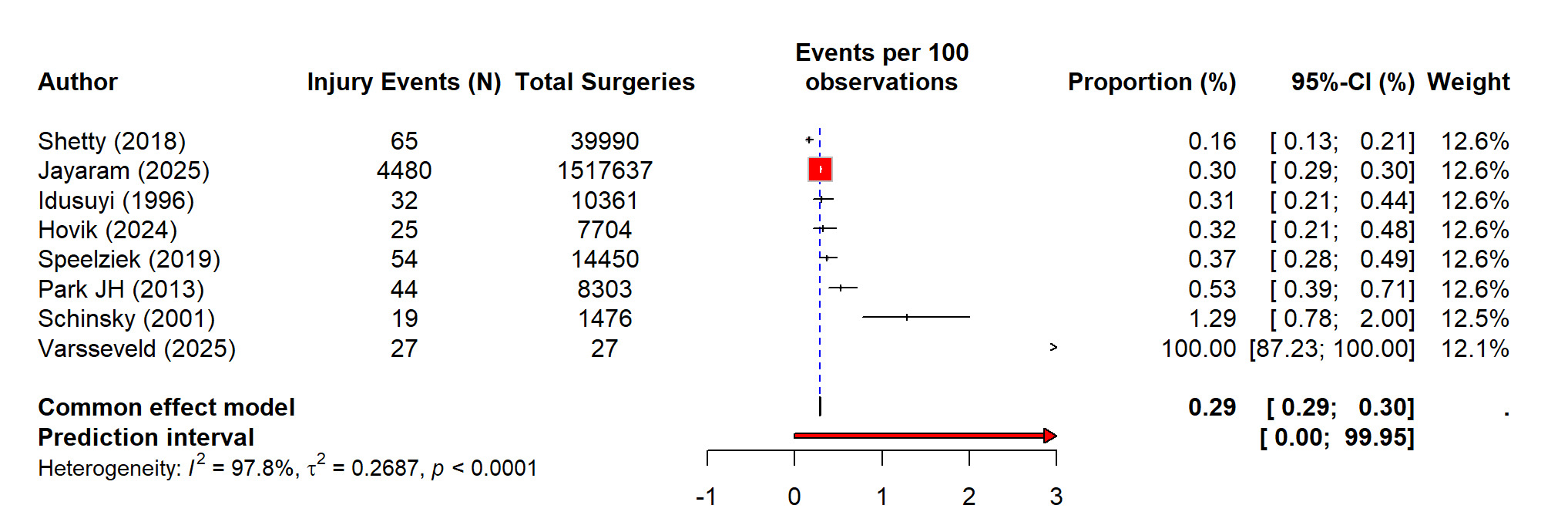
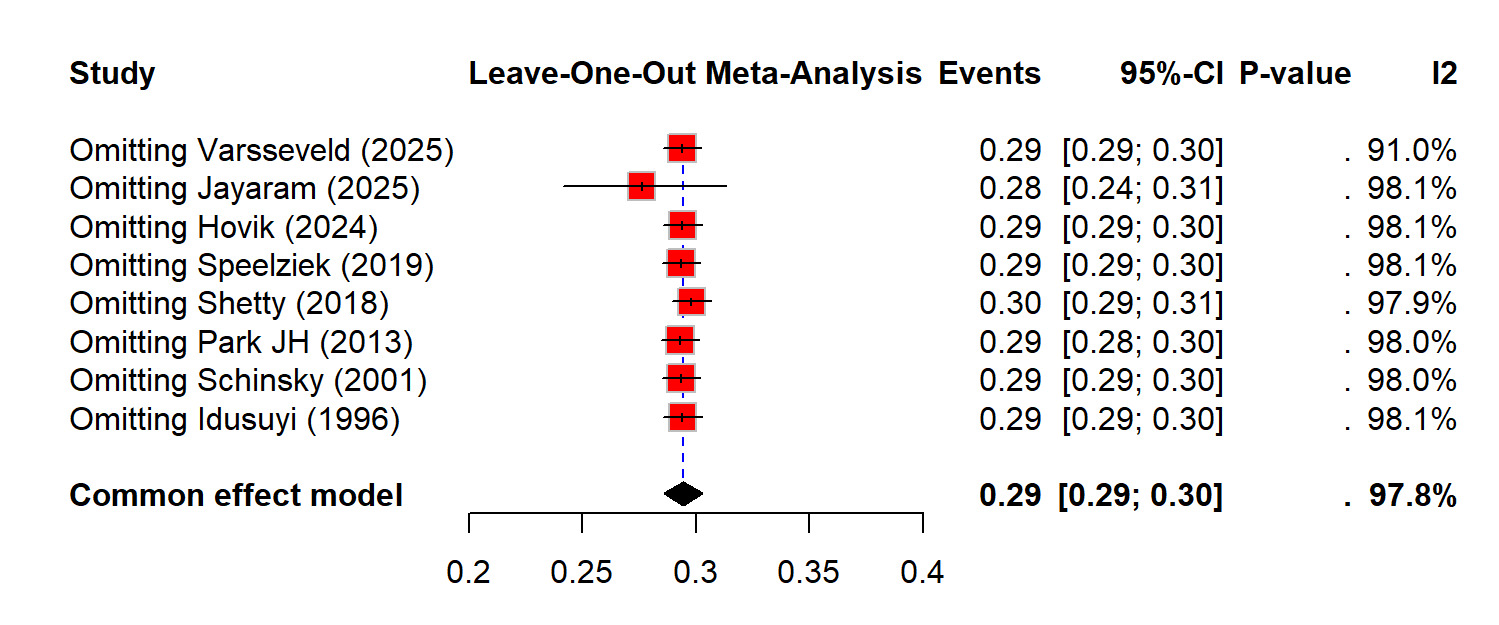
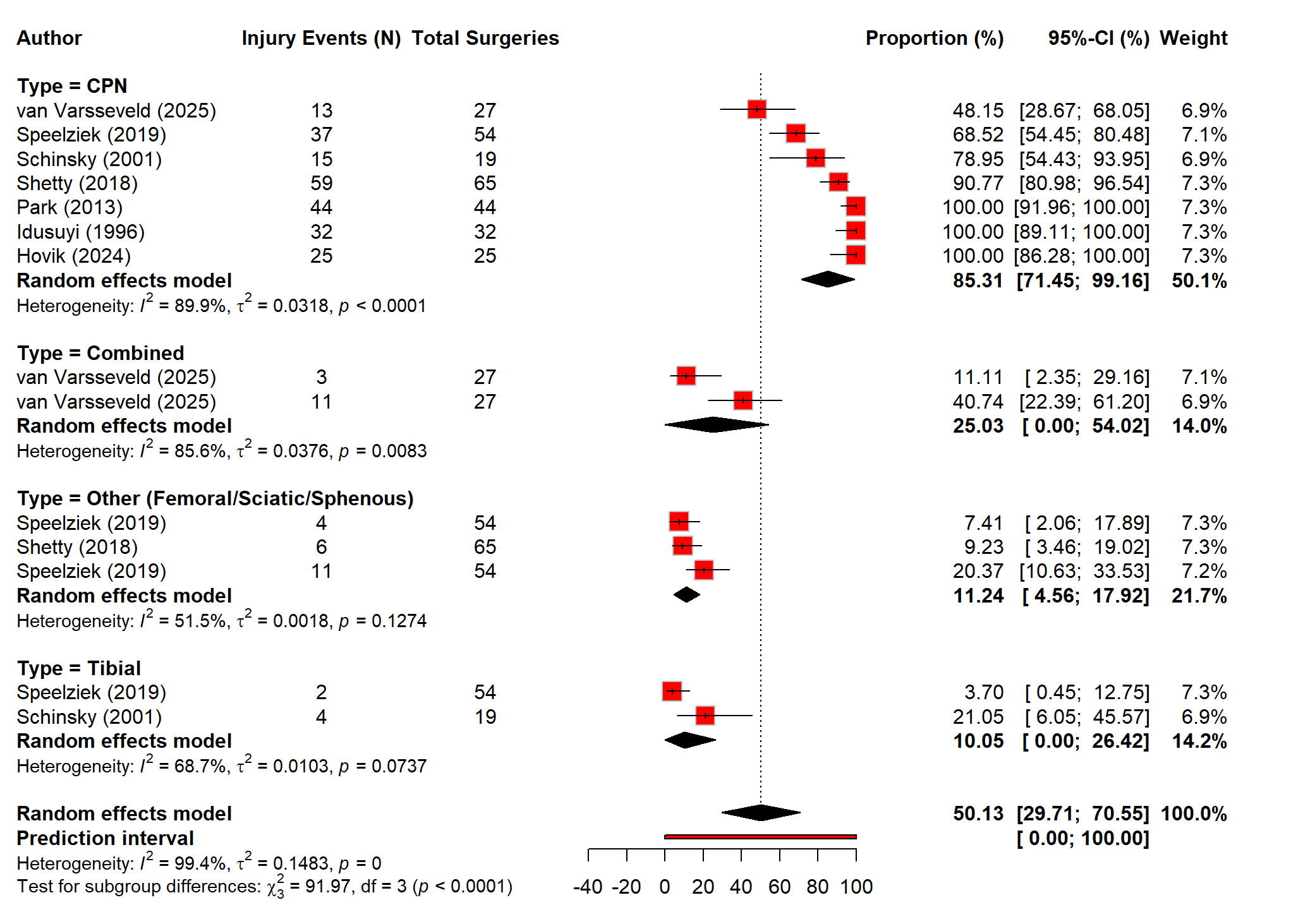
The pooled analysis of eight studies showed that out of 1,599,948 primary and revision TKAs there were 4,746 nerve injury events recorded, and it showed that there is a highly variable incidence of postoperative nerve injury. The random-effects model showed that the rate of nerve injuries after TKA was 0.29% (0.29 to 0.30%) which showed that the burden of nerve injuries after TKA was very rare. However, the heterogeneity was extremely high (I² = 97.8%), which showed that there was inconsistent incidence reporting across studies (Figure 4). The sensitivity analysis demonstrated that removing individual studies did not meaningfully change the overall estimate, supporting the robustness of the pooled result (Figure 5). Out of total nerve injury events, the subgroup analysis based on the nerve type showed that common peroneal nerve (CPN) injuries accounted for the majority of cases (85%), whereas tibial and other nerves, including femoral, sciatic, and saphenous, contributed far fewer injuries (10% and 11% respectively). The combined nerve injuries were also common (25%). These difference in the reported incidence of nerve injuries are signifying statistically (p<0.01) (Figure 6).
Notably, meta regression was performed to explore the significant predictors of the nerve injury after TKA. The demographic parameters like age and gender didn’t impact the incidence of nerve injuries (p > 0.05). Moreover, BMI showed no association with nerve injury risk (p = 0.9883). The presence of comorbidities did not influence the incidence (p = 0.7006), and preoperative deformity also showed no effect (p = 0.9608). Similarly, the type of surgery such as primary versus revision was also not a significant predictor (p = 0.9618) (Table S2). Moreover, the procedure-related factors showed only tourniquet use was the significant predictor of the nerve injury after TKA (p = 0.0136). This indicated that there is a meaningful association with increased risk of nerve injury after TKA. Furthermore, the peripheral nerve block use was not significant (p = 0.2222), and preoperative deformity also showed no significant effect (p = 0.2094). The experience of the surgeon trended toward the significance but did not reach statistical threshold (p = 0.0792) (Table S3).
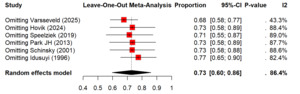
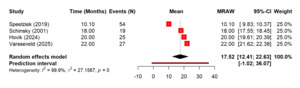
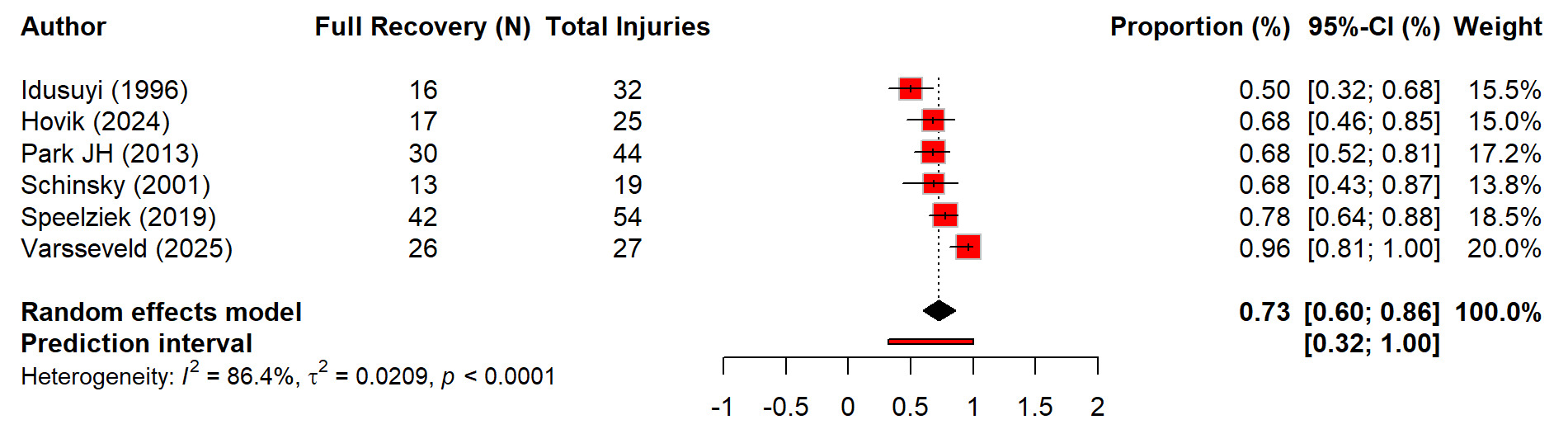
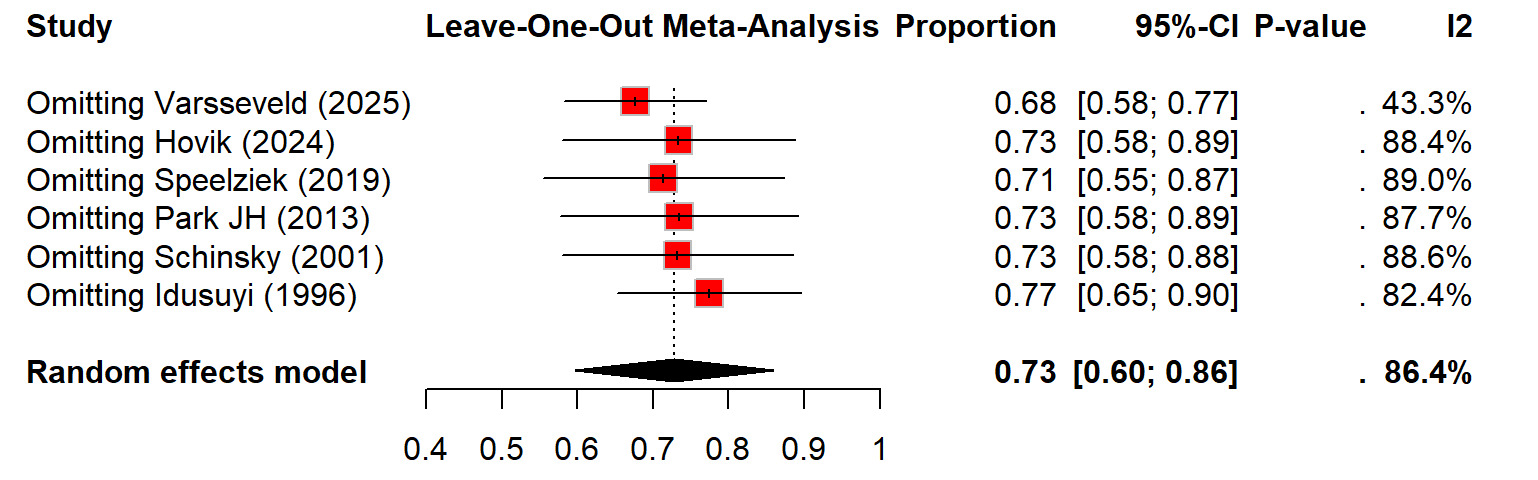
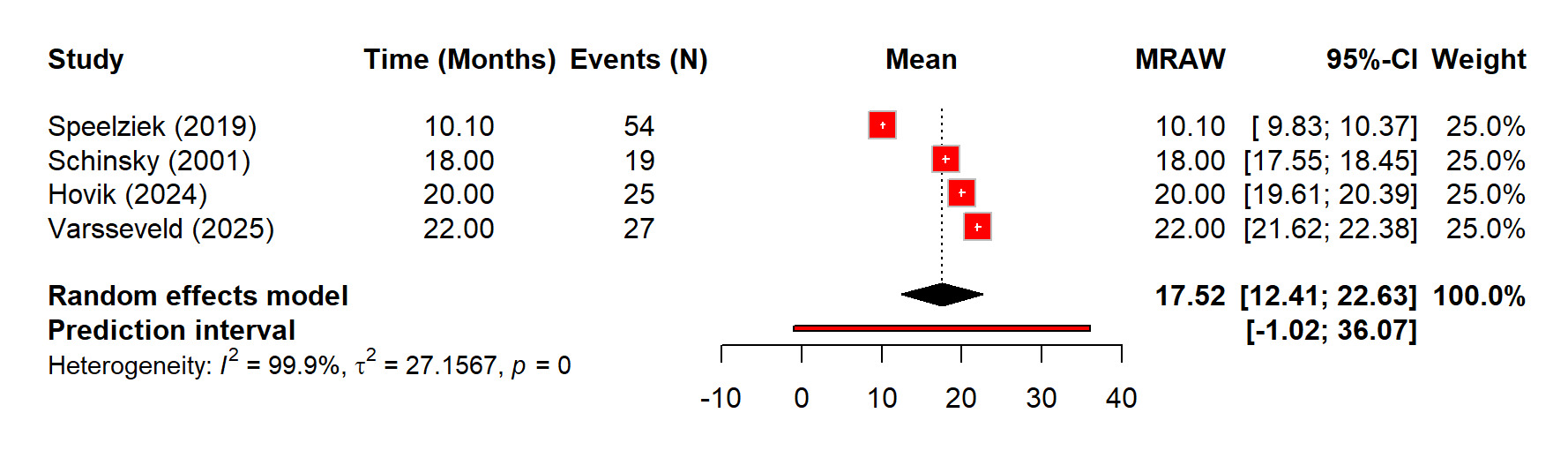
Regarding the recovery after nerve injury, there is overall high recovery rate of 73% (60%–86%) after TKA related nerve injury. Although recovery after nerve injury following TKA was generally favorable, but the heterogeneity was substantial (I² = 86.4%) (Figure 7). The sensitivity analysis showed that removing any single study did not significantly change the pooled estimate, with leave-one-out recovery proportions ranged from 0.68 to 0.77 (Figure 8). The time for full recovery of nerve injury ranged from approximately 10 to 22 months with the mean recovery time of 17.52 months (12.41–22.63), though heterogeneity was very high (I² = 99.9%) (Figure 9).
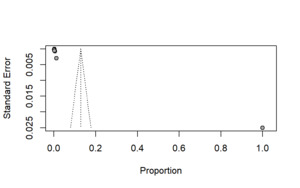
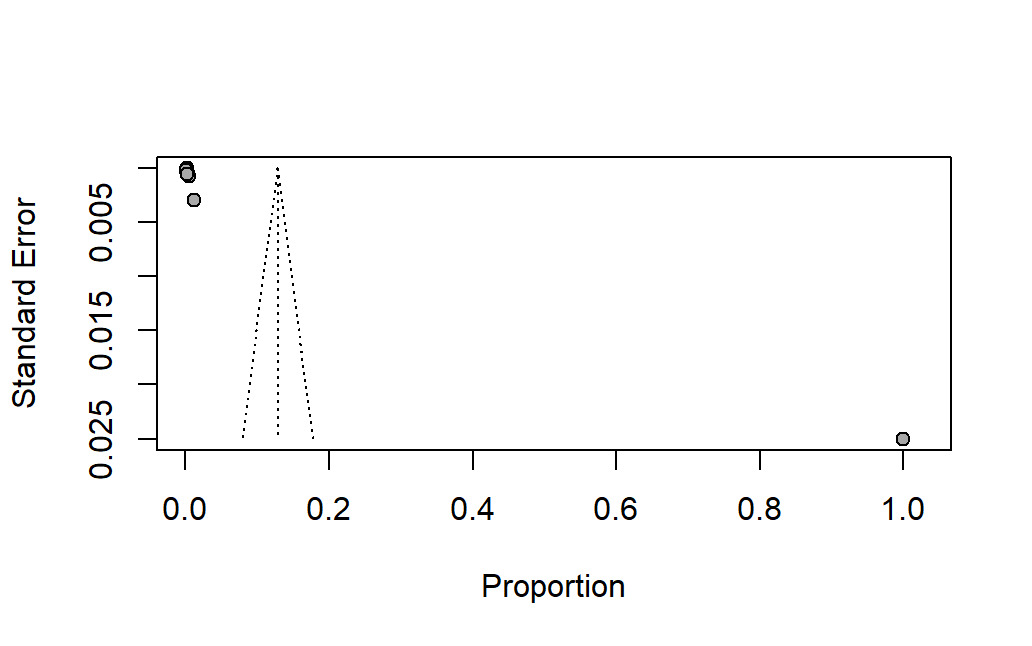
Figure 10 shows the assessment of the publication bias. This shows that there is an asymmetric distribution of studies, which suggested the potential small-study effects. Most of the studies cluster toward the lower end of the proportion axis with the very small standard errors, while there is one study with a very high proportion appears as an outlier on the far right. This imbalance indicates that the available evidence may not be evenly distributed across the full range of effect sizes. The absence of studies in the lower right portion of the funnel further supports the possible publication or reporting bias.
Discussion
Total knee arthroplasty is a widely used procedure for the management of the advanced knee disease. This procedure helps the patients to regain their mobility and reduce the chronic pain.17 Although it is a generally safe procedure, but it can be complicated by several postoperative nerve injuries. These injuries typically involve the common peroneal or the saphenous nerves which may arise from the surgical traction, pressure, tourniquet application, bleeding, or the pre-existing neuropathy.18 This may lead to the symptoms such as the numbness, burning pain, weakness, or even foot drop.19 This systematic review aim to determine the incidence, risk factors, and the recovery patterns of nerve injuries after the total knee arthroplasty.
Notably, this study showed that the pooled incidence of nerve injury following TKA was 0.29%. This rate of injury is aligned with the previous study by Christ et al. (2019) which showed that out of 383,060 TKA cases the rate of nerve injuries was 0.12%.20 The individual studies which included in this review reported the incidence rate of post TKA nerve injuries ranged from 0.16% to 1.3%, which aligns with the previous literature which showed the similar rates. There is a study by Younis et al. (2025) which showed that nerve palsy is a rare but debilitating complication of total knee arthroplasty (TKA), and the reported incidence of these injuries ranged from 0.3% to 10%.21 Moreover, there is a high heterogeneity in our pooled estimate which likely reflects the variations in diagnostic criteria, postoperative follow-up durations, and reporting of specific neurological deficits. Additionally, underreporting of the minor sensory neuropathies in large database studies may contribute to the wide variability.
Furthermore, as reported in the prior orthopedic and neuromuscular literature, the common peroneal nerve (CPN) accounted for the majority of nerve injuries after TKA. In our study, isolated CPN injuries constituted 85% of all cases, which was far more than tibial, femoral, sciatic, saphenous, or the combined neuropathies or injuries. This reason for the common involvement of CPN aligned with the fact that the it traverses around the fibular neck within a tight fascial tunnel. Its course makes it vulnerable to traction, compression, and the stretch during valgus correction. Similarly, a study by Hohmann et al. (2021) shows that the common peroneal nerve (CPN) can be injured during fibular-based posterolateral reconstructions due to its close relationship to the neck of the fibula.22 Bhattacharjee et al. (2025) shows that the mechanism of CPN injury is thought to be via direct stretching of the nerve or indirect traction (i.e., via stretching of the surrounding soft tissue), which narrows both the extra-neural and intra-neural microvasculature and results in impaired blood flow of the CPN.23 Regarding the tibial nerve injury after TKA, a case report by Wang et al. (2021) mentioned that it is very uncommon with rate of 3.7%.24 Moreover, the timing of onset showed two notable patterns with most injuries appeared immediately or within the first few postoperative days after procedure which is consistent with traction or compression mechanisms of injury (Ward et al. 2013).25 Moreover, the delayed-onset neuropathies which included saphenous/IPBSN injuries highlight the potential role of postoperative scarring, entrapment, or implant-related irritation which is aligned with Saif et al. (2020).26
Notably, one of the objectives of this study was to determine which patient or procedural factors increase nerve injury risk. The meta-regression model in our study demonstrated that demographic parameters which included age, sex, BMI, comorbidities, or preoperative deformity did not significantly predict nerve injury. These findings challenged the earlier assumptions from literature which suggested that severe valgus deformity and the diabetes increase risk of nerve injuries. Shetty et al. (2018) showed that risk factors for nerve injury after a total knee arthroplasty (TKA) include revision surgery, female sex, younger age, and certain anatomical and medical conditions like a valgus deformity or prior spinal issues. Furthermore, the intraoperative factors such as longer tourniquet time, longer surgery duration, and excessive blood loss also increase the risk of nerve injuries.27 While Shetty et al. (2018) indicated the higher neuropathy rates in patients with valgus deformity, our pooled analysis found no statistically significant association (p = 0.9608).
One of the important findings of this study is that tourniquet use emerged as the only significant procedural predictor (p = 0.0136). The prolonged use of tourniquet inflation may contribute to ischemic insult, neuropraxia, or delayed conduction block. This aligns with classical orthopedic literature describing tourniquet-related nerve dysfunction as a time-dependent risk. Robinet et al. (3013) shows that nerve injury was attributed to the compression of the tourniquet, as other causes of iatrogenic femoral nerve injury.28 Despite evolving anesthesia and surgical protocols, our findings reinforce caution in high-pressure or extended-duration tourniquet use.
Peripheral nerve blocks, although suspected as the potential contributors, were not associated with nerve injury (p = 0.2222). This is consistent with evidence from anesthesia literature by Tallawy et al. (2025) which showed extremely low rates of persistent neurological deficits after regional blocks.29
The surgeon experience demonstrated a trend toward significance (p = 0.0792), which suggested that high-volume centers may mitigate risk through refined technique and early identification of intraoperative tension. Although not statistically significant, this observation aligned with the surgical outcomes literature which showed that experience as a predictor of technical complications or risk of nerve injury (Shetty et al. 2019).30
Notably, the management of injuries was primarily conservative, which was followed by the standard neuropathy guidelines. The common measures included loosening dressings, reducing traction, and keeping the knee flexed to minimize nerve tension. Surgery was reserved for the definite compression, with the hematoma evacuation and selective decompression showing benefit. The delayed procedures highlighted emerging awareness of the entrapment neuropathies. Rehabilitation such as ankle–foot orthoses and physiotherapy also very essential, while electrical stimulation and neuropathic pain treatments provided additional support when needed (Bhandari et al. 2019).31
The recovery outcomes were generally favorable, with a pooled recovery rate of 73%. This aligns with the classical recovery data which indicated that most neuropraxias resolve within 6–12 months. However, variability in recovery duration (10–22 months) underscores differences in severity and nerve type. The peroneal nerve injuries which are complete palsies, showed longer recovery times, consistent with prior reports. Approximately one-third of the patients experienced persistent deficits. This emphasized that there is the need for early recognition and aggressive functional support to mitigate long-term disability.
Limitations
There are several limitations of this study. There is high heterogeneity in incidence estimates of the included studies, which is likely due to the differences in study design, diagnostic criteria, follow-up duration, and the reporting of motor versus sensory deficits. There are many studies which relied on retrospective data or the administrative coding, which may underestimate minor neuropathies. The procedural details were inconsistently reported, which limited the strength of moderator analyses. Geographic representation was also narrow, with most data originating from the United States. The publication bias was evident, which suggested potential underreporting of negative or low-incidence findings.
Implications and Future Directions
There is a need for the rise in awareness of the nerve injury risk during TKA, especially regarding the modifiable factors such as tourniquet use. The clinicians should emphasize the early recognition, structured postoperative monitoring, and the timely rehabilitation to optimize recovery. There is a need of the standardized diagnostic criteria to ensure consistent reporting across centers. The studies in future should prioritize well-designed prospective studies with uniform definitions, detailed procedural documentation, and longer follow-up.
Conclusion
This study shows that nerve injury after TKA, although uncommon, remains a clinically significant complication, with the common peroneal nerve being the most frequently affected nerve. The use of the tourniquet emerged as the only consistent procedural risk factor, while the demographic and clinical characteristics showed no clear association. Most of the patients achieved meaningful recovery, however a subset of patients experienced the persistent deficits. These findings highlight the importance of the early detection, careful intraoperative technique, and structured rehabilitation.
Abbreviations
-
TKA – Total Knee Arthroplasty
-
CPN – Common Peroneal Nerve
-
IPBSN – Infrapatellar branch of the saphenous nerve
-
PNB – Peripheral Nerve Block
-
AFO – Ankle–Foot Orthosis
-
EMG – Electromyography
-
ROM – Range of Motion
Corresponding Author Details
Abdulmajed A. Alwarafi
Riyadh, Saudi Arabia
Abdulmajeed.ali77@gmail.com