Introduction
Osteoporosis is a skeletal disorder characterized by decreased bone mineral density (BMD) and increased bone fragility, manifested by osteoporotic fractures.1 Common sites of osteoporotic fractures include the wrist, hip, and vertebrae.2 Epidemiological investigations have found that hip and vertebral fractures have high morbidity and mortality rates, imposing a significant economic and societal burden on patients.3 The lifetime risk of osteoporotic fractures is notably high, ranging from 40–50% in women and 13–22% in men.4 Therefore, identifying critical predictors for osteoporotic fracture care and control is essential.
Previous guidelines for the assessment and treatment of osteoporosis have been established, and interventions should be considered for patients who have sustained fragility fractures.5 Osteoporosis in males represents a pathological condition until osteoporotic fracture.6 It is common for patients to suffer an osteoporotic fracture without knowing it, with common clinical manifestations including height loss and thoracic kyphosis; A growing concern is that patients aged ≥50 have a higher risk of sustaining a subsequent fracture within the following 12 to 24 months.7,8 For males, the mortality rate within two years after hip fracture approaches 40%.9 There is, therefore, a gap pertaining to these under- or untreated male patients, and significant advances in diagnostic methods and treatment options are required.10
Bone mineral density (BMD) is a major predictor of osteoporosis, and can be determined using dual-energy X-ray absorptiometry (DXA), a non-invasive diagnostic method. Poor BMD leads to increased mortality11 and a loss of quality of life in people with osteoporotic fractures,12,13 even more so than many common cancers, with the exception of lung cancer.14 The relative risk of fracture increases gradually with decreasing BMD; however, fracture risk does not increase uniformly across populations, especially among males, which contributes to low screening rates and ongoing controversies regarding BMD testing strategies.15,16 However, the association between the number of osteoporotic fractures and BMD needs to be elucidated. Therefore, we analyzed data from the 2005–2010, 2013–2014, and 2017– March 2020 cycles of the National Health and Nutrition Examination Survey (NHANES) database to investigate the association between BMD and the number of osteoporotic fractures and to characterize potential nonlinear patterns and heterogeneity across sex and age groups. We hypothesized that higher femoral neck BMD would be associated with a lower burden of osteoporotic fractures and that this association would differ by sex and age.
Materials and methods
Study population
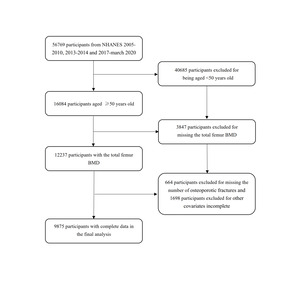
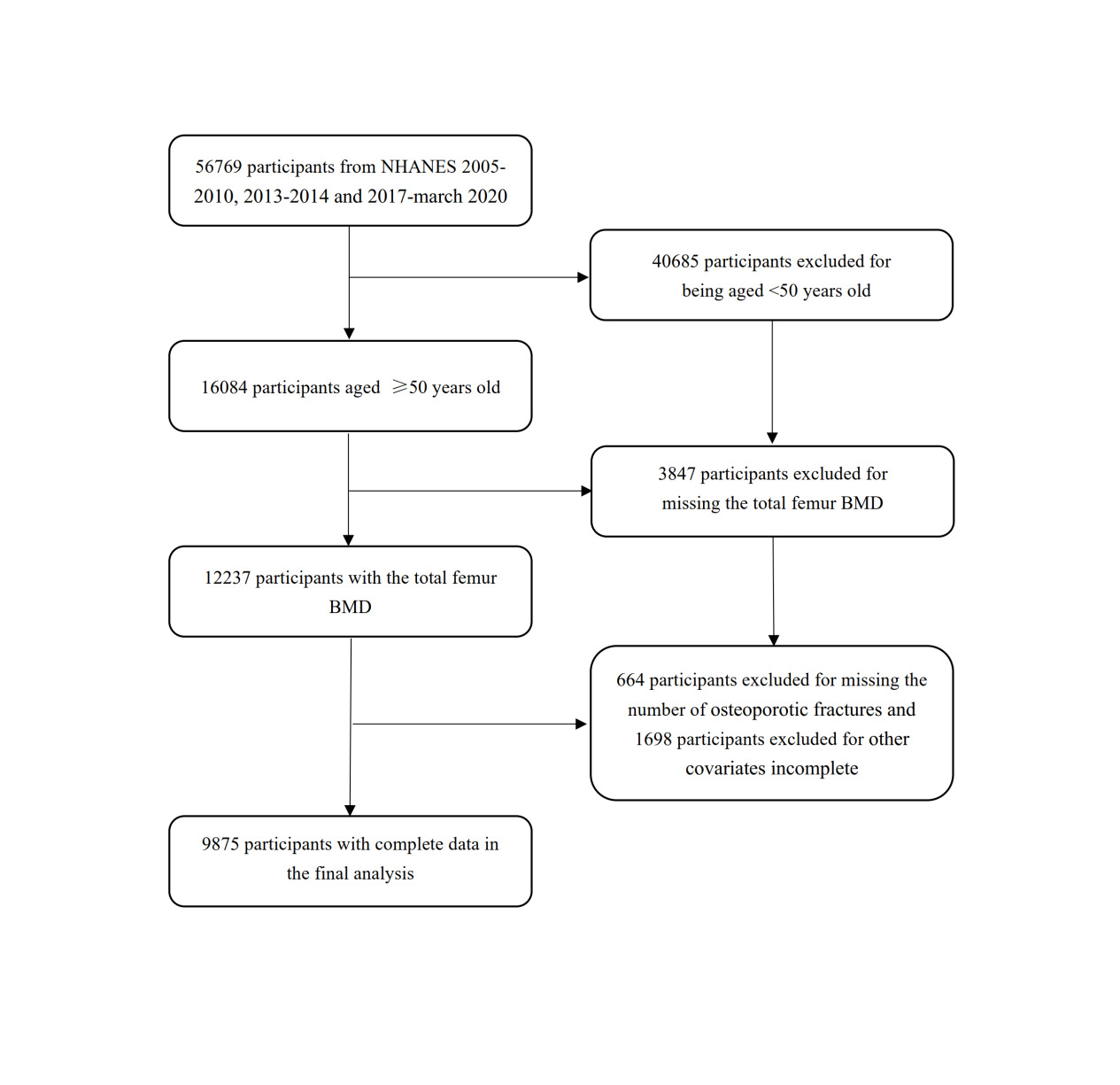
The NHANES database is a national survey which provides information on the nutrition and health of the American population, and is approved by the NCHS Institutional Review Board. The NHANES database is available publicly at www.cdc.gov/nchs/nhanes. We collected data in five NHANES cycles (2005–2010, 2013–2014, and 2017–March 2020). A total of 56769 participants were included. After screening participants aged ≥ 50 years (n = 16084) and excluding those without BMD data (n = 3847) and osteoporotic fracture count data (n = 664), 9875 participants were finally included in our analysis Figure 1.
BMD was measured in units of g/cm² using DXA according to standardized NHANES protocols. DXA examinations were performed using Hologic densitometers, with calibration and quality assurance procedures implemented across survey cycles to ensure measurement comparability. Only valid femoral neck DXA measurements, as defined by NHANES quality control criteria, were included in the analysis. Participants with missing or invalid DXA scans, including those flagged for poor scan quality or technical artifacts, were excluded.
Osteoporotic Fracture history
Osteoporotic fracture history was obtained from the NHANES medical conditions questionnaire and was self-reported. Fracture outcomes were defined using site-specific questions assessing prior fractures of the hip, wrist, and spine. Participants reporting fractures resulting from low-energy mechanisms, including falls from standing height or lower, were classified as having fragility fractures, whereas fractures attributed to high-energy trauma (e.g., motor vehicle accidents or severe injuries) were excluded when such information was available.
Other covariates
For each participant, osteoporotic fracture counts were calculated by summing the number of self-reported fragility fractures across the hip, wrist, and spine sites. Site-specific fracture counts were also derived for secondary analyses. Because vertebral fractures are frequently asymptomatic and may be underreported, fracture counts in this study may underestimate the true burden of vertebral fractures. The following covariates were included in the study: demographic data (sex, age, race/ethnicity, and ratio of family income to poverty); body mass index (BMI); high-density lipoprotein (HDL); glycohemoglobin; alcohol consumption status; diabetes status; physical activity status; smoking status; and osteoporosis-related medication use (including medications for osteoporosis or prevention of bone loss).
Covariates were selected a priori based on established associations with bone mineral density and fracture risk, as well as the availability and consistency of corresponding variables across NHANES survey cycles. Several fracture risk factors incorporated in the FRAX tool, including glucocorticoid dose, rheumatoid arthritis, parental hip fracture, falls history, secondary osteoporosis, vitamin D status, renal disease, and sex hormone levels, were not included due to limited availability or inconsistent measurement in NHANES.
Alkaline phosphatase, phosphorus, and total calcium were evaluated as baseline characteristics but were not included in the fully adjusted regression models. The detailed acquisition process and measurement methods for each variable are available at www.cdc.gov/nchs/nhanes
Statistical analysis
All analyses followed NHANES guidelines for complex survey data and accounted for the complex sampling design using DXA subsample weights, multi-cycle survey weights, and the corresponding strata (SDMVSTRA) and primary sampling unit (SDMVPSU) variables. Continuous variables were summarized as survey-weighted means with 95% confidence intervals (CIs), and categorical variables as survey-weighted proportions. Participants with missing or invalid DXA measurements were excluded based on NHANES quality control criteria.
Osteoporotic fracture outcomes were treated as count variables. Survey-weighted negative binomial regression models were used to estimate incidence rate ratios (IRRs) and 95% CIs for the association between femoral neck bone mineral density (FN-BMD) and fracture counts, given evidence of overdispersion. Three models were specified: an unadjusted model; a partially adjusted model including age, sex, and race/ethnicity; and a fully adjusted model further incorporating socioeconomic status, body mass index, lipid profile, glycohemoglobin, lifestyle factors, diabetes status, and osteoporosis-related medication use. Prespecified adjustment sets were defined based on a causal framework; collinearity was assessed and no problematic multicollinearity was detected. Missing data were handled using a complete-case approach, which may introduce residual confounding.
Restricted cubic spline (RCS) functions within survey-weighted negative binomial models were applied to evaluate potential nonlinear associations between FN-BMD and fracture counts, with model fit and nonlinearity assessed using information criteria and formal tests. Sex- and age-stratified analyses examined effect modification. Sensitivity analyses included redefining outcomes as any fragility fracture (binary), restricting counts to the first fracture per site, excluding uncertain or high-energy mechanisms, and considering time since fracture where available. A two-sided P value <0.05 was considered statistically significant. All analyses were performed using R (version 4.2.0) and EmpowerStats (version 5.0).
Results
Baseline characteristics
A total of 9,875 participants aged ≥50 years were included in the final analysis, representing a nationally representative sample of U.S. adults. The weighted mean age was 62.91 years (95% CI: 62.57–63.25), with 48.77% males and 51.23% females. Most participants were non-Hispanic White (77.58%). The weighted mean body mass index was 28.64 kg/m² (95% CI: 28.47–28.80), and the mean femoral neck bone mineral density (FN-BMD) was 0.76 g/cm² (95% CI: 0.76–0.77). Overall, 4.68% of participants reported a history of osteoporotic fracture, and 14.36% had diabetes. Detailed baseline characteristics are shown in Table 1.
Model diagnostics and selection
Fracture outcomes were modeled as count variables. Diagnostic comparisons between survey-weighted Poisson and negative binomial regression models demonstrated substantial overdispersion under the Poisson assumption. Residual-versus-fitted plots showed markedly improved residual distributions with negative binomial models, supporting their use in primary analyses (Figure S1).
Overall association between femoral neck BMD and fracture counts
In survey-weighted negative binomial regression analyses, higher FN-BMD was consistently associated with lower osteoporotic fracture counts. In fully adjusted models, FN-BMD showed a strong inverse association with hip fracture counts (IRR: 0.000, 95% CI: 0–0.005; P < 0.001) and total osteoporotic fracture counts (IRR: 0.015, 95% CI: 0.004–0.053; P < 0.001) (Table 2).
Nonlinear association assessed by restricted cubic splines
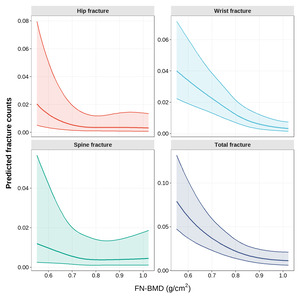
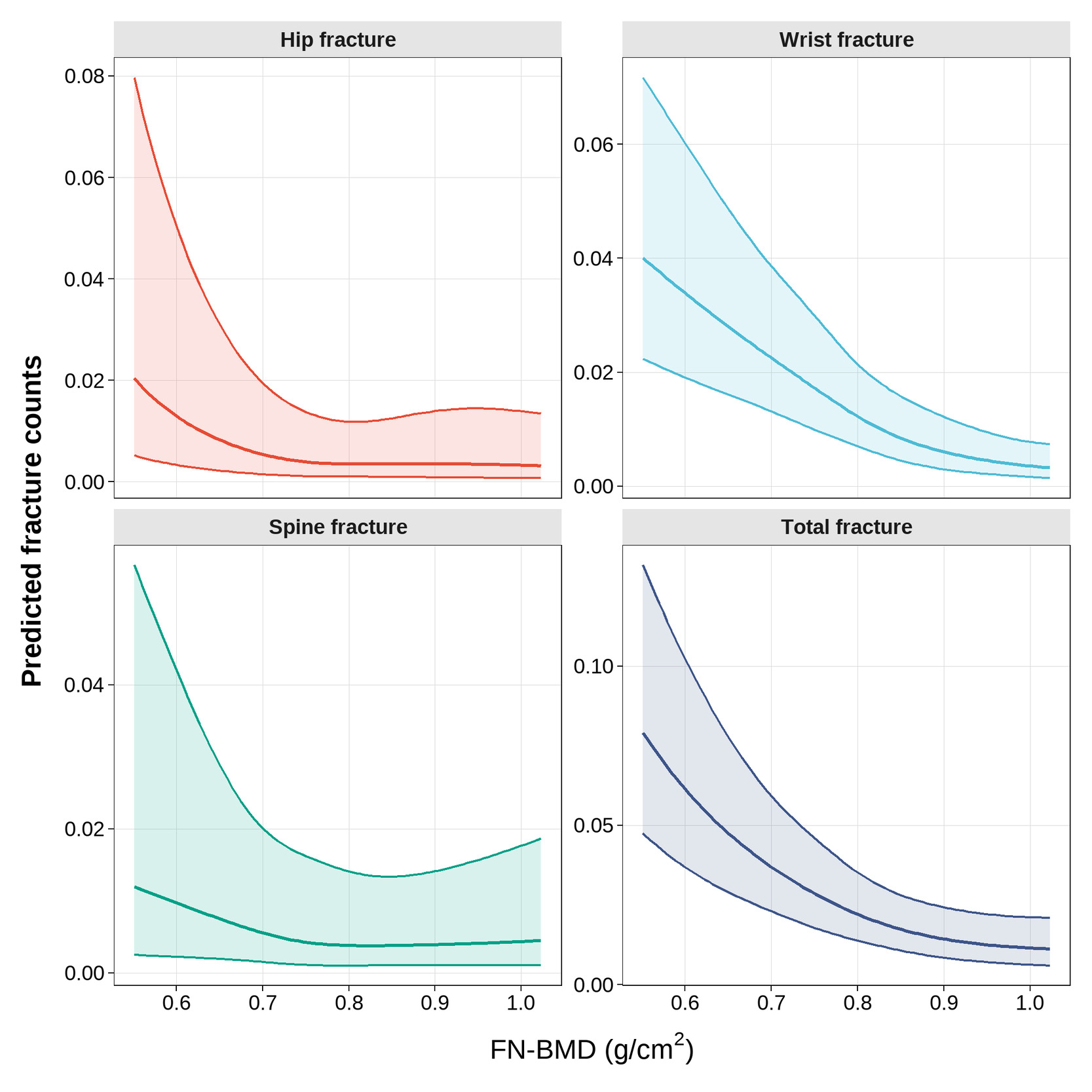
Restricted cubic spline analyses further demonstrated a nonlinear inverse association between FN-BMD and predicted fracture counts. Across hip, wrist, spine, and total osteoporotic fractures, fracture counts decreased progressively with increasing FN-BMD, with a steeper decline observed at lower BMD levels and a gradual attenuation at higher BMD ranges (Figure 2). Model fit statistics and formal tests for nonlinearity supported the use of spline-based models (Table S1).
Sex- and age-stratified analyses
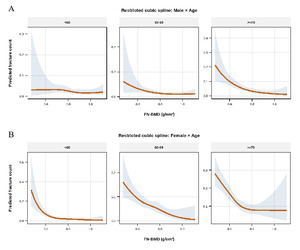
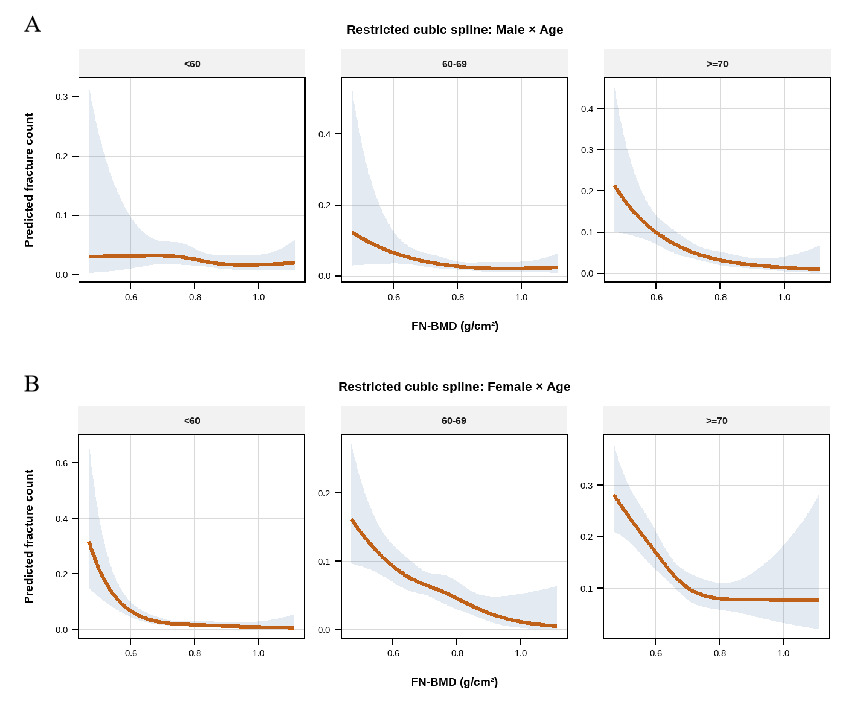
Sex- and age-stratified restricted cubic spline analyses demonstrated substantial heterogeneity in the association between femoral neck bone mineral density (FN-BMD) and predicted total osteoporotic fracture counts (Figure 3). Among males, a pronounced inverse and nonlinear association between FN-BMD and fracture counts was observed in participants aged ≥70 years, with predicted fracture counts decreasing sharply at lower FN-BMD levels and gradually leveling off at higher BMD ranges. In contrast, the inverse association was weaker and more gradual among males aged 60–69 years, while no clear association was evident among males aged <60 years. Among females, higher FN-BMD was consistently associated with lower predicted total osteoporotic fracture counts across all age groups. The inverse association was strongest among females aged <60 years and ≥70 years, whereas a more gradual decline in fracture counts was observed among females aged 60–69 years. Notably, the magnitude and shape of the association differed between sexes within corresponding age strata.
Sex-stratified regression analyses further supported these patterns, showing an overall inverse association between FN-BMD and fracture counts in both males and females, with effect sizes varying by sex (Table S2). Age-stratified analyses indicated stronger associations in older age groups compared with younger participants (Table S3). Consistent results were observed in joint sex- and age-stratified models (Table S4).
Sensitivity analyses
Sensitivity analyses using alternative fracture definitions and outcome specifications yielded results consistent with the primary analyses. The direction and magnitude of the association between FN-BMD and fracture outcomes remained stable across multiple sensitivity scenarios, supporting the robustness of the findings (Table S5).
Discussion
In this cross-sectional study, we used the NHANES 2005–2010, 2013–2014, and 2017–March 2020 databases to analyze the association between the number of osteoporotic fractures and femoral neck BMD in adults. We found an overall inverse association between the number of osteoporotic fractures and femoral neck BMD in the hip, wrist, and total fracture sections among all participants. Moreover, we observed potentially nonlinear patterns in the association between the number of osteoporotic fractures and femur neck BMD among the different subgroups, with heterogeneity by sex.
Since 1994, osteoporosis diagnosis guidelines produced by the World Health Organization (WHO) were based on a T-score of −2.5 or lower for BMD assessed by DXA at the femoral neck or spine.17 The T-score, therefore, has always been used as a critical reference for the diagnosis and treatment of osteoporosis. However, it is not regarded as an absolute fracture risk18,19; numerous studies have demonstrated that a single measurement of BMD is not sufficient to assess fracture risk because many fragility fractures occur in people who do not reach the threshold as defined by the WHO criteria.20,21 Other factors, independent of BMD, have been identified, including a history of fracture, glucocorticoid use, family history of fracture, smoking, excessive alcohol consumption, and low BMI.8,22 To better estimate fracture risk, the WHO Fracture Risk Assessment Tool (FRAX)23–26 is now used worldwide, which can predict the patient’s future risk in the next 5–10 years.27 The advantage of FRAX is that it is not completely dependent on BMD, but the disadvantage is that it does not account for dose responses for several risk factors (e.g., glucocorticoid exposure, cigarette smoking, alcohol intake), history of falls, and different intervention thresholds.22 Although BMD is widely used in fracture risk assessment, it should be interpreted as a continuous risk indicator rather than a strict intervention threshold. The upper assessment threshold is generally higher than intervention thresholds because almost no individuals will be at risk of reclassification when BMD is added to FRAX; in the UK, the upper assessment threshold was set at 1.2 times that of the intervention threshold.28 Regardless, BMD alone is not the optimum intervention threshold for various reasons, but it is still the most commonly used and robust predictor of fracture risk.
Osteoporosis in men is always underappreciated, underdiagnosed, and undertreated, in contrast to postmenopausal women.29 The main points of contention are the age and frequency of BMD screening and the different reference criteria for BMD testing. There is consensus that screening for osteoporosis with BMD testing in women aged ≥ 65 and in men aged ≥ 70, For men aged 50 to 70 and postmenopausal women under 65, common recommendations suggest BMD testing should be conducted if there are fracture risks (e.g., fracture after age > 50, height loss of more than 4 cm, low body weight, hypogonadism, hyperthyroidism, drugs affecting bone metabolism, lifestyle factors . et al.).30–32 However, the US Preventive Services Task Force (USPSTF) thinks there is insufficient evidence to assess the balance of benefits and harms of screening BMD in men; also, no specific age was recommended.33 In fully adjusted analyses, higher femoral neck BMD was associated with lower fracture burden, particularly for hip and total osteoporotic fractures, while associations with vertebral fractures were weaker. The weak association with vertebral fracture may be related to the test femur neck BMD rather than test spinal BMD,34,35 and many factors limit the availability of reliable information on spine fracture history.36 In sex- and age-stratified analyses, stronger inverse associations were observed among older males (≥70 years), whereas associations were weaker or absent in younger age groups. Our data found that men aged 60–70 have higher activity levels compared to those aged ≥70; additionally, NHANES data may have limitations in reliably capturing spine fractures. These findings suggest that the the association between BMD to fracture burden varies by age and sex, and may be less pronounced in younger males compared with other fracture risk factors. In females, age ≥ 70 is a strong predictor for all fracture models, and age <60 is another strong predictor for hip fractures. The association between BMD and fracture counts in females demonstrated nonlinear patterns across age groups, which may be influenced by factors such as bone geometry, hormonal changes, fall risk, and sex-specific bone loss trajectories,37 leading to earlier onset of osteoporosis, and fracture occurrence compared with males.31 Overall, these findings indicate that the observed associations between BMD and fracture burden differ across sex and age strata, and should be interpreted in the context of other demographic and clinical factors.
The International Osteoporosis Foundation (IOF) recommends selecting the femoral neck as the BMD test site and T-scores standardized measurements using the NHANES III reference base for Caucasian women aged 20–29 years38; Previous studies suggest that there is no significant difference in the gradient of risk between men and women for any of these fracture outcomes. Marshall et al., in a meta-analysis,39 reported hip fracture risk increases 2.6-fold (95% CI 2.0–3.5) for a 1 standard deviation (SD) decrease in hip BMD; any fracture risk increases 1.5-fold (95% CI 1.4–1.6) for a 1 SD decrease in any site BMD. Another meta-analysis of data from 12 cohort studies showed that the risk gradient for any fracture increased with age, and the gradient of risk was lower than for hip fractures, similar in both sexes; absolute risks may differ.40 Specifically, the gradient of hip fracture risk decreased with age, but the absolute risk still increased. Others have noted significant sex differences in young men, which became less significant in older men.41,42 In the present study, fracture burden was assessed using fracture counts rather than fracture incidence, which does not directly reflect absolute fracture risk. Nevertheless, fracture counts decreased progressively with increasing femoral neck BMD, and the observed patterns support a continuous, inverse association rather than discrete fracture thresholds. These findings suggest that BMD should be interpreted in conjunction with demographic and clinical factors rather than as a sole determinant of fracture risk.
To our knowledge, ours is the first study to evaluate the association between BMD and the number of osteoporotic fractures based on the NHANES database, which uses a large sample size, includes both sex- and age-stratified analyses, and adjusts an adequate number of covariates in survey-weighted regression models. However, this study has some limitations. First, owing to the cross-sectional design of the NHANES database, the age at the time of fracture did not completely match the BMD measurement time. We evaluated the association between BMD and the number of osteoporotic fractures and cannot infer causality or temporal relationships between exposure and outcome. Therefore, further prospective studies are necessary. Second, the information on osteoporotic fractures from participants was self-reported, and many fractures, particularly vertebral fractures, may be underreported. Residual confounding cannot be excluded, particularly for fracture risk factors not captured or incompletely measured in NHANES, such as glucocorticoid exposure, rheumatoid arthritis, falls history, vitamin D status, renal disease, and sex hormone levels. Third, treatment management during different periods may have impacted BMD. Finally, there may have been recall bias in the questionnaires, possibly impacting the covariates. Considering the cross-sectional design of this study, further prospective and long-term studies are worth conducting to verify our findings.
Conclusion
We found a significant inverse association between femoral neck BMD and the number of osteoporotic fractures in U.S. adults aged ≥50 years, with substantial variation by sex and age. Nonlinear patterns were observed in the association between femoral neck BMD and fracture counts; however, these patterns should be interpreted as continuous, observational risk gradients rather than discrete inflection points or fracture thresholds. The magnitude and shape of the association differed between males and females, with stronger inverse associations observed in older age groups, showing more consistent patterns among females and greater heterogeneity among males. In contrast, the association between femoral neck BMD and fracture counts was weak or absent in males aged <60 years, indicating that the observed association between BMD and fracture burden is limited in this subgroup and should be interpreted cautiously in the context of other clinical risk factors, rather than as a standalone determinant of fracture risk.
Acknowledgements
Not applicable.
Funding
This study was supported by Special project on Kunshan Municipal Science and Technology Bureau (KS2343).
Data Availability Statement
The NHANES database is available publicly at www.cdc.gov/nchs/nhanes.
Conflict of Interest
The authors declare that they have no competing interests.
Ethical Statement
Not applicable.
Author Contributions
Yunfeng Wan: methodology, software, validation, writing-original draft preparation. Xuyang Sheng: formal analysis, investigation. Wenqing Kan: visualization. Jun Ji: resources, data curation. Hongkou Xu: writing-review and editing, supervision. Jianhua Hu: conceptualization, validation, project administration, funding acquisition. Wenqing Lu: conceptualization, validation, project administration, funding acquisition.
All authors have read and agreed to the published version of the manuscript.