Introduction
Calciphylaxis (also termed calcific uremic arteriolopathy) is a rare and serious medical condition characterized by abnormal, excessive calcification in the underlying vascular system of the subcutaneous fat and dermal tissues.1 This calcification leads to clinically significant manifestations such as painful ischemic sores, often with associated tactile hyperesthesia.1 Whereas calciphylaxis is most commonly associated with uremia and end-stage renal disease (ESRD), other characteristics and comorbid conditions have been postulated to be related, including female sex, White race, obesity, diabetes, protein undernourishment, coagulopathy, use of a vitamin K inhibitor such as warfarin, and endocrine or metabolic derangements such as hyperparathyroidism and hypercalcemia.1–5 Most patients with non-nephrogenic calciphylaxis have more than one of these conditions.6 In the US, the incidence of calciphylaxis in patients with ESRD requiring hemodialysis has been reported at 35 cases per 10,000 persons.7 Diagnosis carries a 6-month survival rate of 57%.3
The resultant clinical effects of calciphylaxis create perioperative treatment challenges. Skin ulcerations can predispose to infection with the potential for spread to deeper sites, and death from sepsis is a common finding in calciphylaxis vasculopathy.2 A particularly debilitating feature of this disease is pain. The pain associated with ischemic calciphylaxis lesions is described as excruciating and unrelenting and often leads to a decline in patients’ functional status and to repeated hospitalizations.8 For patients with severe cases without adequate pain control, poor quality of life is expected. Surgical debridement of necrotic ulcers is often necessary to preserve surrounding tissue, and patients may require repeated surgical procedures after diagnosis and near the end of life.3,4,9
Currently, no standard treatment protocol exists for calciphylaxis nor are comprehensive guidelines available for perioperative care of patients with calciphylaxis. The purpose of this review was to evaluate existing literature in order to offer guidance on treating patients with this challenging disease through the perioperative period.
Methods
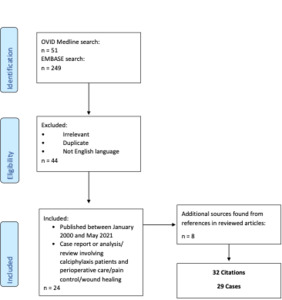
We conducted a narrative review of OVID Medline and EMBASE databases from January 2000 to May 2021 for English-language–only articles by using the search terms “calciphylaxis,” “calcific uremic arteriolopathy,” “anesthesia,” “anesthesiology,” “perioperative,” “acute pain,” “pain management,” and “postoperative pain.” Additional sources were also identified from cited references in the articles initially returned by the search.
The articles were organized according to 4 categories: 1) case reports/case series with an emphasis on surgical/procedural treatment, 2) case reports/case series with an emphasis on pain-control measures, 3) case reports/case series with an emphasis on medical management, and 4) retrospective analyses and reviews evaluating risk factors, treatment outcomes, and general recommendations (Tables 1-5). For each identified case, data for patient age, sex, and comorbid conditions were collected and summarized along with author recommendations for management (if available). For each identified retrospective analysis or review article with general conclusions for disease and management recommendations, the level of supporting evidence was evaluated and a descriptive summary of each was generated.
Table 1.Summary of Case Reports/Case Series Focusing on Surgical Procedures
| First author, year |
Age, sex |
Surgery/procedure |
Comorbid conditions |
Recommendations/conclusions |
| Adler, 202110 |
56, male |
Partial penectomy |
ESRD, diabetes, hypertension |
Penectomy may confer infectious source control and improve patient quality of life in penile calciphylaxis with a high risk of progression. |
| Diasty, 202111 |
39, female |
Mitral valve replacement and tricuspid valve repair |
Posttransplant ESRD, hyperparathyroidism, hypertension, anemia, mitral stenosis, tricuspid regurgitation |
Exposure to calciphylaxis risk factors should be minimized during surgery for high-risk patients. |
| Horishita, 200412 |
43, male |
Left lower-extremity amputation |
ESRD |
Laser Doppler flowmetry is a useful tool for monitoring peripheral blood flow to prevent intraoperative ischemia. |
| Kriskovich, 200013 |
53, female |
Subtotal parathyroidectomy |
ESRD |
Early diagnosis and surgical intervention may be beneficial for survival. Parathyroidectomy may be safely performed in this patient population without increased complication rates. |
| Kriskovich, 200013 |
77, female |
Subtotal parathyroidectomy |
ESRD, diabetes, hypertension |
See above. |
| Kriskovich, 200013 |
53, male |
Subtotal parathyroidectomy |
ESRD |
See above. |
| Kriskovich, 200013 |
30, female |
Total parathyroidectomy |
ESRD |
See above. |
| Kriskovich, 200013 |
58, female |
Subtotal parathyroidectomy |
ESRD |
See above. |
| Naik, 200414 |
43, female |
Surgical debridement, total thyroidectomy and parathyroidectomy |
ESRD, diabetes, morbid obesity, hyperparathyroidism |
Aggressive wound debridement and excision of necrotic tissue is warranted to prevent disease progression. |
| Naik, 200414 |
50, female |
Surgical debridement, excision of necrotic fat tissue |
ESRD, diabetes, morbid obesity |
See above. |
| Naik, 200414 |
70, male |
Surgical debridement, subtotal parathyroidectomy |
ESRD, diabetes, morbid obesity, coronary artery disease, hyperparathyroidism |
See above. |
Abbreviation: ESRD, end-stage renal disease.
Table 2.Summary of Case Reports/Case Series Focusing on Pain-Control Measures
| First author, year |
Age, sex |
Procedure |
Comorbid conditions |
Recommendations/conclusions |
| Edquist, 202015 |
48, male |
Bilateral cryoneurolysis of pudendal and sciatic nerves |
ESRD |
Image-guided neurolysis may be used to treat refractory pain as an alternative to amputation. |
| Green, 200016 |
51, male |
Neurolytic lumbar sympathetic block |
ESRD |
Lumbar sympathetic blockade should be considered for pain management. |
| Mach, 202117 |
78, male |
Ultrasound-guided dorsal penile nerve block with 4% phenol |
ESRD, diabetes, heart failure |
US-guided phenol nerve blocks may be used for long-term targeted neurolysis to manage refractory pain. |
| Polizzotto, 200618 |
54, female |
Subcutaneous ketamine infusion (500 mg), sufentanil (900 mcg) |
ESRD, obesity, hypertension, anxiety |
An opioid, benzodiazepine, and ketamine combination was most successful in pain control. In the choice of an opioid, renal failure status should be considered. |
| Polizzotto, 200618 |
65, female |
Subcutaneous fentanyl infusion (300 mcg) with 40 mcg boluses for breakthrough pain, midazolam (2.5 mg) |
ESRD, bipolar affective disorder, Parkinson disease, diabetes, obesity, ischemic heart disease |
See above. |
| Polizzotto, 200618 |
59, female |
Subcutaneous infusion of sufentanil (80 mcg), midazolam (15 mg), haloperidol (2.5 mg), subcutaneous ketamine (50 mg) |
ESRD, diabetes, obesity, hypertension, ischemic heart disease, alcohol dependence |
See above. |
| Sato, 200119 |
50, female |
Morphine and epidural nerve block |
ESRD, hyperparathyroidism |
Morphine and epidural nerve block were unsuccessful in controlling pain. Early diagnosis and avoidance of precipitating factors, such as corticosteroids, are important. |
Abbreviation: ESRD, end-stage renal disease.
Table 3.Summary of Case Reports/Case Series Focusing on Medical Management
| First author, year |
Age, sex |
Treatment |
Comorbid conditions |
Recommendations/conclusions |
| Biswas, 201620 |
62, female |
Prednisone, 40 mg/d, tapered to 25 mg/d; risedronate, 70 mg/wk; vitamin K, 2 mg/d |
Psoriatic arthritis, dyslipidemia, fibromyalgia, GERD, peptic ulcer disease, hypothyroidism |
In a patient with nonuremic calciphylaxis, systemic prednisone treatment showed improvement in wound healing and pain control. The article’s author acknowledged other research showing an association between corticosteroid use and disease onset in other patients. |
| Cole, 202021 |
56, female |
Twice weekly topical oxygen therapy at home |
ESRD, diabetes, amyloidosis, anemia, atrial fibrillation, coronary artery disease, hypertension, hyperlipidemia |
Topical oxygen therapy resulted in complete ulcer healing at 9 weeks. |
| El-Taji, 202022 |
59, male |
Conservative |
Posttransplant ESRD, diabetes, dilated cardiomyopathy, peripheral vascular disease, atrial fibrillation, congestive heart failure |
Conservative treatment may be successful in cases without wet gangrene or significant pain. |
| Lintzeris, 201823 |
62, female |
Purified collagen matrix with polyhexamethylene biguanide topical therapy |
ESRD, diabetes |
PCMP topical therapy resulted in successful ulcer healing at 11 weeks after treatment. |
| Starcea, 201824 |
4, female |
Oxygen therapy, CPAP, inotropic therapy, anticonvulsants |
ESRD attributable to posterior urethral valve |
Prophylaxis of hyperphosphatemia and secondary hyperparathyroidism and early diagnosis of calciphylaxis may improve survival. |
| Yalin, 201325 |
67, male |
Ceftazidime and neutramycin for sepsis |
ESRD, diabetes, hyperparathyroidism, hypoalbuminemia |
Increased awareness and early recognition of calciphylaxis with subsequent infection precaution may aid in improving prognosis of the disease. |
| Yalin, 201325 |
74, female |
Imipenem plus cilastatin for sepsis |
ESRD, chronic pyelonephritis, hyperparathyroidism, hypoalbuminemia |
See above. |
| Yalin, 201325 |
42, male |
Calcium acetate phosphorus-binder, vancomycin, fluconazole for sepsis and candidiasis |
ESRD, hypertension, hyperparathyroidism, hypoalbuminemia |
See above. |
| Yalin, 201325 |
57, male |
Antibiotic therapy for sepsis, aluminum hydroxide for hyperphosphatemia |
ESRD, diabetes, hyperparathyroidism, hypoalbuminemia |
See above. |
| Yalin, 201325 |
48, male |
Pulse steroid and cyclophosphamide therapy |
ESRD, diabetes, hypocalcemia, hyperphosphatemia |
See above. |
| Yalin, 201325 |
42, female |
Vancomycin and neutramycin for sepsis |
ESRD, diabetes, hypertension |
See above. |
Abbreviations: CPAP, continuous positive airway pressure; ESRD, end-stage renal disease; GERD, gastroesophageal reflux disease; PCMP, polyhexamethylene biguanide.
Table 4.Summary of Retrospective Analyses and Guidelines/Recommendations Relevant to Perioperative Management
| First author, year |
Study description |
Author recommendations/conclusions |
Level of evidence |
| Brandenburg, 201926 |
Open-label single-arm trial involving 14 patients with calciphylaxis who received intravenous SNF472 3 times per week for 12 weeks during hemodialysis sessions |
The treatment was well tolerated and wound healing, pain control, and quality of life improved. |
IV |
| Chang, 20192 |
Narrative review describing calciphylaxis characteristics, diagnosis, and management strategies |
Recommendations relevant to perioperative care:
- Warfarin discontinued, replaced with apixaban
- Surgical debridement and hyperbaric oxygen therapy for wound healing
- Avoid use of morphine in patients with renal failure; replace with fentanyl and methadone (no active metabolites that accumulate in patients with renal failure)
- Sodium thiosulfate treatment for wound healing may also have benefits for pain control.
|
VI |
| Chinnadurai, 202027 |
Survey of 106 UK clinicians specializing in palliative and renal medicine that evaluated trends in pain management of calciphylaxis and gave clinical practice recommendations based on the results |
Authors recommended the following:
- Referral to palliative medicine, wound care specialists
- Continuous subcutaneous infusion of alfentanil for pain control, dosage adjusted according to pain relief and neurotoxicity
- Morphine, oxycodone, tramadol, and codeine to be avoided because of neurotoxicity in renal failure patients
- Gabapentin (100 mg) for neuropathic pain
- Stepwise approach for procedural analgesia beginning with short-acting fentanyl, progressing to subcutaneous fentanyl, sedation with midazolam, and general anesthesia with propofol and fentanyl
|
VII |
| Darres, 201928 |
Retrospective analysis of 71 patients with calciphylaxis receiving sodium thiosulfate therapy who were also treated with topical cerium nitrate-silver sulfadiazine |
Topical cerium nitrate-silver sulfadiazine was shown to be potentially protective because of vascular decalcification, reactive oxygen species chelation, and infection prevention. The authors recommended a prospective comparative trial to confirm results. |
V |
| Garza-Mayers, 201829 |
Retrospective analysis of 20 patients with ESRD and calciphylaxis who were treated with apixaban for DVT or AF |
No adverse effects were reported. The authors suggested apixaban as a safe and effective alternative to warfarin in this patient population. |
V |
| Gould, 202130 |
Development of a modified version of the Bates-Jensen Wound Assessment Tool for evaluation of calcific uremic arteriolopathy (calciphylaxis) wounds specifically |
BWAT-CUA includes 8 items for evaluation of calciphylaxis wounds: 1) necrotic tissue type, 2) necrotic tissue amount, 3) exudate type, 4) exudate amount, 5) skin color surrounding wound, 6) peripheral tissue edema, 7) peripheral tissue induration, and 8) granulation tissue. A standardized wound assessment scale may be used for more accurate comparisons between patients in both clinical practice and in clinical research studies. |
VI |
| Maida, 202031 |
Two older women (85 and 69) diagnosed with nonuremic calciphylaxis presenting as lower-leg ulcers were treated with topical cannabis-based medicines |
Topical cannabis-based medicines were well tolerated, provided good pain control, and promoted complete wound closure. |
VI |
| Nigwekar, 20187 |
Matched case-control study of 62 patients that reported ORs of variables previously identified as being associated with calciphylaxis |
A positive association was found between calciphylaxis and hypercalcemia, hypoalbuminemia, calcitriol use, and warfarin use. The authors also tentatively identified a negative association between calciphylaxis and statin use. |
IV |
| Olaniran, 201932 |
Retrospective analysis of data of 24 inpatients diagnosed with calciphylaxis who died as of April 30, 2018, designed to evaluate use of palliative care consultations and end-of-life care measures |
Palliative care teams were consulted for only half of the patients identified, and many received intensive life-prolonging measures during a terminal admission, suggesting the need for better integration of palliative care services—especially considering the high mortality and morbidity associated with calciphylaxis. |
VI |
Abbreviations: AF, atrial fibrillation; BWAT-CUA, Bates-Jensen Wound Assessment Tool for Calcific Uremic Arteriolopathy; CUA, calcific uremic arteriolopathy; DVT, deep vein thrombosis; ESRD, end-stage renal disease.
Table 5.Summary of Retrospective Analyses and Reviews of Survival, Risk Factors, and Treatment Outcomes of Patients With Calciphylaxis
| First author, year |
Study description |
Author recommendations/conclusions |
Level of evidence |
| Kramann, 201333 |
Primary study of skin specimens from 7 patients with calciphylaxis that focused on pathogenesis and histology |
Calciphylaxis pathogenesis involves upregulation of BMP-2 signaling, hydroxyapatite deposition, and matrix remodeling in the subcutaneous tissue. |
VI |
| Lal, 20099 |
Retrospective analysis of cases of 26 patients with biopsy-proven calciphylaxis, evaluating risk factors, parathyroidectomy, and surgical debridement |
Surgical debridement showed statistically significant improvement in survival; parathyroidectomy did not. The authors suggested that a multidisciplinary approach to treatment with early diagnosis, aggressive medical management, surgical debridement, and parathyroidectomy is warranted for these patients. |
V |
| McCarthy, 20163 |
Retrospective analysis of 101 patients with calciphylaxis seen at Mayo Clinic in Rochester, Minnesota, between January 1, 1999, and September 30, 2014, that evaluated risk factors, comorbid conditions, and treatment response |
Results showed that surgical wound debridement and parathyroidectomy in patients with hyperparathyroidism improved survival. Tissue plasminogen activator, sodium thiosulfate, and hyperbaric oxygen therapy may also be considered for wound healing and seem to be well tolerated with no adverse effects. |
V |
| Robert, 202034 |
Retrospective analysis of 8 patients with calciphylaxis treated by Rheopheresis after sodium thiosulfate and discontinuation of vitamin K antagonists failed to resolve symptoms |
Of the 8 patients, 5 had complete remission of disease after Rheopheresis. The authors recommended a prospective study with a larger sample to confirm benefits of the Rheopheresis approach. |
VI |
| Torregrosa, 201535 |
Combined retrospective analysis of cases of 12 patients and a prospective analysis of cases of 11 patients, all of whom received bisphosphonate therapy for calciphylaxis |
Intravenous bisphosphonate therapy may be useful for treatment of calciphylaxis wounds despite potential for exacerbation of bone disease in renal failure patients. |
V |
| Weenig, 20074 |
Retrospective analysis including 64 patients with calciphylaxis that evaluated risk factors and prognosis |
Calciphylaxis is a multifactorial disease with high mortality that is associated with obesity, systemic corticosteroid use, liver disease, increased serum aluminum, and increased erythrocyte sedimentation rate. Of note, warfarin use and increased calcium-phosphate product were not reliably associated with calciphylaxis development. Surgical debridement was shown to improve survival but not parathyroidectomy. The authors recommended modification or avoidance of identified risk factors as a means for primary or secondary prevention of the disease. |
V |
| Zakher, 202136 |
Retrospective analysis of 10 patients with calciphylaxis that evaluated treatment outcomes involving sodium thiosulfate and intensified hemodialysis therapy |
Mortality rates improved and wounds resolved after multimodal treatment that included sodium thiosulfate and intensified hemodialysis (>20 h/wk). |
VI |
Abbreviation: BMP, bone morphogenetic protein
Results
The database search identified 300 potentially relevant articles. A total of 29 case reports from 32 sources met criteria for inclusion (Figure). As described in Methods, Tables 1 through 5 summarize the reports by subject area: surgical/procedural treatment; pain control measures; medical management; relevant retrospective analyses and recommendations relevant to perioperative management; and risk factors, treatment outcomes, and general recommendations.
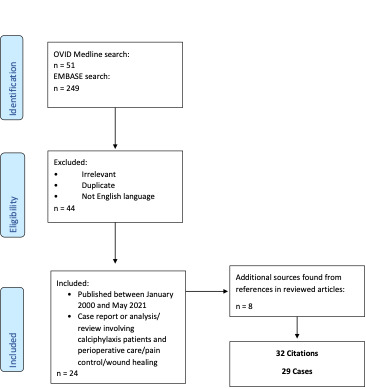
Figure 1.
Although a standard protocol does not exist for treating calciphylaxis, the literature search identified several common therapies being used including intravenous sodium thiosulfate,3,6,36,37 parathyroidectomy,3,9,13 surgical debridement,2,4,9,14 bisophosphonate,35 cinacalcet,5 and hyperbaric oxygen therapy.3,5,21 However, the reports conflicted regarding the efficacy of each, which will be described in detail in the Discussion. There are also no comprehensive guidelines for perioperative management of patients with calciphylaxis. A retrospective analysis of cases of 5 patients treated with parathyroidectomy reported no specific additional risks of surgery.13 Several reports showed that ultrasound-guided neurolytic therapy with cryoneurolysis15 or phenol injection16,17 was effective for pain relief. Interestingly, one report documented a case of postoperative calciphylaxis in a patient with ESRD who was exposed to multiple calciphylaxis-associated risk factors perioperatively.11 Thus, it may be prudent to identify at-risk patients undergoing surgery and control risk factor exposure to minimize development of calciphylaxis postoperatively.
Discussion
Recommendations for Perioperative Management
Based on the literature review and case reports, we recommend the following perioperative management for patients with calciphylaxis (Supplemental Table).
Preoperative
Preoperative evaluation should assess for common comorbid conditions including ESRD, diabetes, obesity, and hyperparathyroidism and/or hypercalcemia.1–5 A thorough history and physical examination should be conducted before surgery with special consideration to baseline pain scores and functional activity because of the debilitating effects of calciphylaxis on patient pain and functional capacity.32 For these assessments, structured questionnaires have been reported as more accurate than subjective assessments.38 For patients taking long-acting opioids for pain management, we recommend having them take their extended-release medication the morning of surgery. Given the large proportion of patients with calciphylaxis who are dialysis dependent, timing dialysis must be considered when scheduling surgery—optimally 24 hours after dialysis to minimize the impact on volume status and electrolyte balance (level II).18 Laboratory studies should be obtained to assess for relevant parameters including estimated glomerular filtration rate, electrolyte imbalances, serum glucose, and anemia. ESRD, diabetes, and obesity are associated with increased perioperative risk for cardiovascular events. The following validated models may be useful for predicting cardiac complications: revised cardiac risk index39 and American College of Surgeons surgical risk calculator.40
Intraoperative
To our knowledge, no intraoperative issues specific to calciphylaxis require special consideration. A retrospective analysis of 5 patients with calciphylaxis undergoing parathyroidectomy showed no additional surgical risks.13 However, given the cutaneous involvement of the disease, care should be taken when positioning the patient to avoid exacerbating existing wounds. As with other patients with ESRD, fluid and electrolyte status need to be monitored preoperatively and intraoperatively to avoid complications such as pulmonary edema, anesthesia-induced hypotension, and arrhythmias attributable to hyperkalemia or hypocalcemia (level II).41 For patients undergoing parathyroidectomy to treat hyperparathyroidism, parathyroid hormone assays should be considered to monitor any decrease in parathyroid hormone levels during surgery.42
Pain control should be aggressively pursued using a multimodal approach. Because of the prevalence of renal impairment in patients with calciphylaxis, medications such as nonsteroidal anti-inflammatory drugs (which impair renal autoregulation) or morphine (risk of metabolite accumulation) may be contraindicated.43 Therapies used successfully preoperatively should be continued perioperatively, eg, fentanyl/fentanyl analogs, benzodiazepines, local anesthetics such as lidocaine, and neuropathic pain agents such as anticonvulsants or ketamine (level IV).18 Low-dose ketamine infusions have been used in opioid-tolerant patients for acute pain control without adverse effects or need for dose modification due to renal impairment and therefore may be an adjunctive therapy for achieving pain control (level IV).44 Intraoperative ketamine at lower rates (0.1-0.5 mg/kg/h) has been used for perioperative pain management of burn patients with similarly complex wounds.38
Calciphylaxis may be triggered or exacerbated by corticosteroids and blood transfusions. Therefore, systemic corticosteroid use should be avoided, and steps should be taken to minimize perioperative blood transfusions (eg, reversing anemia associated with chronic kidney disease through iron and/or erythropoietin supplementation before surgery and careful fluid management and repletion during surgery). A patient with calciphylaxis and POEMS syndrome who received dexamethasone every 2 weeks had remission of both POEMS syndrome and the calciphylaxis skin lesions.37 Dexamethasone may still be an option for postoperative nausea and vomiting prophylaxis.
If complications due to peripheral ischemia (based on location of calciphylaxis lesions) are a concern, laser Doppler blood flowmetry monitoring may be used to maintain peripheral blood flow in conjunction with vasodilators such as prostaglandin E1 analogs (level IV).12
If a patient has diabetes, intraoperative glycemic control is important.
Postoperative
For this study, we identified 3 areas of interest that should be considered in postoperative care.
First, risk factors and triggers need to be avoided. For example, vitamin K antagonists such as warfarin may trigger or exacerbate calciphylaxis.18 Therefore, we recommend giving a direct anticoagulant to patients who require postoperative anticoagulation therapy. A retrospective study of 20 patients with ESRD and calciphylaxis showed that apixaban was a safe and effective alternative to warfarin.29 Other potential calciphylaxis triggers include corticosteroids19,31 and blood transfusions.11
Second, poor wound healing is another postsurgical issue. A retrospective study of 71 patients with calciphylaxis undergoing sodium thiosulfate treatment showed that cerium nitrate-silver sulfadiazine therapy might be useful for vascular decalcification and infection prevention.28 A study evaluating a biofilm of purified collagen matrix with polyhexamethylene biguanide showed complete ulcer healing and re-epithelization in a patient with calciphylaxis.23 Use of the novel therapy SNF472, an intravenous formulation of myoinositol hexaphosphate, to inhibit hydroxyapatite crystal formation and prevent vascular calcification has shown potential therapeutic benefit.26 Hyperbaric oxygen therapy and referral to a burn center are options for managing recalcitrant wounds8,21; however, this therapy—like others for calciphylaxis—has not been validated by adequately powered prospective studies.45
Finally, adequate pain control is notoriously difficult to achieve for patients with calciphylaxis, requiring a multifactorial approach. Reported successful strategies include a combination of fentanyl/fentanyl analogs with benzodiazepines and neuropathic agents such as ketamine (level IV).18,44 Success has also been reported for neurolytic nerve blocks using phenol16,17 or cryoneurolysis.15 Diagnostic nerve blocks using fast-acting agents such as lidocaine or benzocaine may be useful for determining efficacy for individual patients. Given the high rates of morbidity and mortality associated with calciphylaxis, discussing palliative care options with patients early in the treatment process may be useful to maximize quality of life (level V).27,32
Conclusions
Calciphylaxis is a complex disorder affecting those with liver disease, diabetes, obesity, or, more commonly, uremia and ESRD. The formation of calcified microvascular deposits leads to microthrombosis, ischemia, and painful cutaneous manifestations. For these patients, preoperative and intraoperative disease management involves monitoring and optimizing comorbid conditions, taking into account any electrolyte imbalance. In postoperative management, the challenges involve potential triggers of calciphylaxis such as warfarin and corticosteroids. Moreover, poor wound healing and difficult pain control are frequent issues. A multifactorial approach that includes nerve blocks, renal-sparing opioids, benzodiazepines, and/or ketamine is recommended to optimize postoperative pain.
Acknowledgment
Marianne Mallia, ELS, MWC, senior scientific/medical editor, Mayo Clinic, substantively edited the manuscript. The Scientific Publications staff at Mayo Clinic provided proofreading, administrative, and clerical support.
Author Contributions
All authors made substantial contributions to the analysis and interpretation of data for this manuscript. The drafting of the manuscript was performed evenly by all authors, and all authors are accountable for the accuracy and integrity of the work.
Declaration of Competing Interests
The Authors declare that there are not conflicting interests.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.