Introduction
Spinal stenosis is a condition characterized by the narrowing of the spinal canal and subsequent compression of the spinal cord and nerve roots.1,2 Spinal stenosis has a prevalence of 250,000 to 500,000 people in the United States with an expected growth in cases due to an aging population. Only 1% of these cases are reported to occur in the thoracic region.3
Thoracic spinal stenosis (TSS) may co-occur with cervical or lumbar stenosis.4 TSS is most frequently due to ossification of the posterior longitudinal ligament (OPLL), ossification of the ligamentum flavum (OLF), posterior osteophytes, or thoracic disk herniation (TDH). Of these problems, OLF is the most reported causative factor, but a combination of these problems can also be responsible.2,3,5,6
In this case series, we report a unique association among three cases of critical multilevel spinal stenosis caused by diffuse spinal hyperostosis in young patients with morbid obesity.
Case 1
Patient #1 is a morbidly obese, 32-year-old male with a BMI of 58. The patient reported to his primary care physician with progressive, ongoing numbness to his lower extremities beginning in his bilateral feet intermittently. Over the following four months, his numbness progressively worsened with extension up his legs and to the nipple line bilaterally. Prior to imaging, the patient reported significant weakness to his lower extremities causing him to fall multiple times and rendering him wheelchair bound. The patient had nil relevant for past medical, social, and family history.
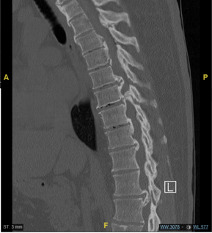
Preoperative CT and MRI images of the spine demonstrated thoracic stenosis from T3–5 and again at T7-8 due to disc herniations and diffuse ossification of ligamentum flavum (Figure 1). Further findings included mild to moderate C4-5 neuroforaminal stenosis secondary to a left paracentral disc herniation, a right sided C5-6 paracentral disc herniation, also abutting the spinal cord and causing central canal stenosis, and a broad-based C6-C7 disc.
On examination at the time of surgery, the patient had Achilles tendon contractures bilaterally. The motor exam demonstrated weakness of hip flexors, 1/5 in the left leg and 3/5 in the right leg. Hoffman’s test was negative bilaterally with down going Babinski bilaterally. Clonus was sustained bilaterally.
To manage the symptomatic thoracic myelopathy, patient #1 underwent a posterior T4-T5 decompression and fusion. Postoperatively, the patient reported improved sensation, notably to the right leg but to a lesser extent to the left leg. He also noticed that the spasms in his legs and hips were improved since surgery.
Case 2
Patient #2 is a morbidly obese, 35-year-old male with a BMI of 41. This patient, with unknown past medical history, initially presented to the hospital with acute onset of difficulty ambulating. He stated a 1-year history of lower back pain and an acute 1-week onset of bilateral lower extremity numbness, weakness, and frequent falls. The patient indicated that his back pain worsened when standing straight and extending his back with improvement when leaning forward.
An MRI revealed severely degenerative changes with disc herniations at T8-9, T12-L1, L4-5, and L5-S1. He additionally had severe central canal stenosis at T8-9 with hyperintense signal along the cord. CT was later obtained and demonstrated severe heterotopic bone formation of the posterior longitudinal ligament along with marked facet hypertrophy at several levels, leading to critical thoracic and lumbar spinal stenosis.
On examination, the patient had difficult ambulating bilateral lower extremity weakness. Hip flexion (L2), ankle dorsiflexion (L4), great toe extension (L5), and ankle plantarflexion (S1) was assessed as 4/5 bilaterally. There was also 4/5 weakness assessed in ankle dorsiflexion of the left leg. The sensory examination was normal in the upper and lower extremities.
He later underwent an L2 laminectomy for decompression of the central canal, lateral recess and foraminal stenosis. The postoperative course was complicated by durotomy, and examination was notable for absent motor function and minimal sensation in his bilateral lower extremities. A repeat MRI showed worsening T7-T9 stenosis due to soft tissue edema and T8 hematoma. Later in hospitalization, he had an elective VIR IVC filter placement with discharge to acute rehabilitation and recovered motor and sensory function by 6 month follow-up.
Case 3
Patient #3 is a 31-year-old female with a BMI of 60 and a past medical history of hypertension. She presented with progressive numbness of the bilateral lower extremities after falling on her knees as she felt her legs give way. She was unable to stand and felt weak and an ambulance was called. Prior to this episode, she had progressive weakness in her lower extremities bilaterally as well as numbness in her feet over several months. In the emergency department, she reported numbness throughout her abdomen and lower extremities.
The patient further reported, since the time of her fall, that she has not urinated. The patient describes she did not feel an urge to urinate and denied urinary incontinence. A straight catheter was performed in the emergency department and 736 cc of urine was recorded, consistent with urinary retention. Another catheter inserted several hours later measured 400 cc.
The patient was awake, alert, and oriented to person, place, and time. The patient reported tenderness to percussion over the thoracic spine with diminished light touch sensation below T4 including loss of perianal sensation. Reflexes were recorded as 2+ bilaterally with negative Hoffman’s, and downgoing Babinski.
A CT scan of the thoracic and lumbar spine demonstrated multilevel spondylosis with extensive posterior marginal osteophytes, as well as ossification of the posterior longitudinal ligament resulting in spinal canal stenosis spanning T1-T8 level, most severe at T7-T8.
MRI of the spine found increased STIR signal within the spinal cord at T5-T6. The imaging and examination findings were consistent with spondylotic myelopathy.
Patient #3 was scheduled for a posterior thoracic decompression with possible thoracic fusion; however, this was cancelled due to active COVID-19 infection. She is currently being managed non-operatively as she elected for non-operative management following her COVID infection.
Discussion
Various syndromes and diseases are implicated in the pathogenesis of spinal stenosis. Congenital stenosis is responsible for only 9% of cases with acquired causes including trauma, degenerative changes, iatrogenic causes, and systemic processes.7 Ossification of spinal ligaments can be idiopathic or secondary to metabolic and genetic disease. For example, prior case reports have been published documenting associations between thoracic spinal stenosis due to ossification secondary to Diffuse Idiopathic Skeletal Hyperostosis (DISH), achondroplasia, skeletal fluorosis and renal osteodystrophy.6,8–10 None of the patients described in this case series had any known medical history which may explain their degree of spinal hyperostosis and ligament ossification.
This case series describes a rare cause of spinal stenosis with similar characteristics between the patients. All three patients were obese and presented with multilevel severe spinal hyperostosis leading to significant cord compression with a variation of spinal cord injury (complete vs incomplete). OLF and OPLL is most frequently seen in the East Asian population, although this case series documents patients of Hispanic and South-East Asian race.11,12
Ossification of the spinal ligaments occurs due to endochondral ossification of hypertrophied fibrous tissue within the ligaments.13–15 The case series demonstrates that degree of obesity, and resulting metabolic changes, may suggest an explanation for the ligament ossification.13 This is supported by several prior studies which found that metabolic factors were significant in DISH.16 In these studies, serum levels of insulin and adipokines, such as leptin, were greatly elevated which subsequently may affect bone metabolism and cause heterotopic bone formation in ligaments.
Patients may suffer from multiple levels of compression, as observed in our patients, and result in a double crush syndrome.17 This occurs when there is distinct compression at two or more locations along the course of a nerve which synergistically increases symptom intensity. For example, the second patient in this series presented with a footdrop and disc herniation. The patient deteriorated after lumbar decompression and was found to have numerous calcified thoracic disc herniations.
Dural defects associated with OLF may complicate surgical management and choice of technique. A prior study of 98 patients with thoracic spinal stenosis due to OLF found dural ossification involved in 26 patients.18 Similarly, another study of 182 patients undergoing decompressive laminectomy and OLF removal found that 52 patients had intraoperative evidence of dural adhesion and 23 patients had dural ossification.19 When this occurs, surgical risks, such as injury to the spinal cord or nerve root and cerebrospinal fluid (CSF) leakage due to dural tears, are increased. This highlights the importance of pre-operative imaging to identify these defects which enable the surgeon to counsel the patient appropriately and anticipate dural laceration during surgery.
Conclusion
While diffuse hyperostosis is an uncommon etiology of spinal stenosis, it is an important condition to recognize due to an association with increased surgical risk and complications. Patients can present with insidious or acute onset of myelopathy, typically affecting the lower extremities and ability to walk. Pre-operative imaging is essential in guiding the surgeon to potential pitfalls of management when performing surgical decompression and removal of the ossified ligaments.
Data Availability
All patient information data is deidentified and it is not available to public.
Author Contributions
All authors conceptualized the project and have approved for submission. DA, GA, KJD, CLM wrote the initial manuscript and performed a review of literature. EOK and AHD revised the manuscript, edited, and supervised.
Funding
No sources of funding for this project
Ethical Approval
No IRB is required for a case series of 3 patients, all patients have provided informed consent to the clinician.
Competing Interests
DA, GA, KJD, CLM have no disclosures to report. EOK receives consulting fees from Seaspine, and Spineart. AHD receives consulting fees from Stryker, Orthofix, Spineart, and EOS, research support from Southern Spine, and Fellowship support from Orthofix.

_and_sagittal__b)_views_-_for_patient__1_demonstrating_di.png)
_and_t2_axial_mri_(right_image)_of_patient__2_revealed_ossification_o.png)



_and_sagittal__b)_views_-_for_patient__1_demonstrating_di.png)
_and_t2_axial_mri_(right_image)_of_patient__2_revealed_ossification_o.png)