Background
Total hip arthroplasty is one of the most successful procedures and it is performed widely in order to restore mobility and to reduce the burden of degenerative joint disease for millions of patients worldwide.1 Over past few decades, multiple technological and clinical advisement have been introduced in order to improve outcomes and turning THA into a reliable cornerstone of modern joint reconstruction.2 Although it is safe but there are some complications which can still occurred after this procedure. Among these, nerve injuries is one of the most common which delay restorations of normal function due to postoperative neurological deficits.
Nerve injuries after THA are uncommon, however their impact can be significant. The incidence these injuries ranged from 0.6 to 3.7%, and it is highest in patients with developmental hip dysplasia and previous hip surgery.3 These clinical presentation of these injuries ranged from mild sensory deficit to serious motor deficits which can alter gait, limit independence and reduce the quality of life.4 Most commonly affected nerves include the sciatic, femoral, pudendal, and obturator nerves.5 Most of these injuries recover with time, while other injuries persist and they lead to long-term disability.
The reported incidence of nerve injury after THA varies widely across studies due to differences in diagnostic criteria, follow up duration and surgical approach. Posterior lateral and anterior approaches have their own patterns of risk which is shaped by exposure technique, traction forces and patient positioning.6 Patient related factors of these injuries include preexisting deformities, previous hip surgery, high body mass index and female gender are often linked to higher susceptibility.7 There are several other factors which are related to surgeons and nature of surgery which included his experience, implant selection and the way tissues are handled during the procedure.8 Despite extensive research in this field, there is still lack of clear understanding about the incidence of nerve injuries and factors most strongly predict these injuries and which preventive strategies provide the most reliable protection.
Moreover, there are several mechanisms which can lead to nerve injuries. These include direct trauma from instruments, compression from retractors, stretching during hip dislocation or reduction thermal injury from cement and postoperative hematoma formation.9 Some injuries are noticed immediately while others appear more gradually which make it very difficult to diagnose them early. Electrophysiological testing and imaging can help with diagnosis but prevention remains the most dependable strategy.10
Because of these uncertainties there is a need of robust high-quality evidence which can accurately define the incidence of nerve injuries after THA and clarifies the major risk factors outcomes and emerging trends. Therefore, a systematic review and meta-analysis is needed in order to address these uncertainties by consolidating data from diverse studies and providing a comprehensive assessment of the burden and clinical significance of postoperative nerve injury. The present study aims to support the evidence based surgical decision making enhance patient counseling and reinforce preventive strategies within total hip arthroplasty.
Materials and Methods
This study was designed as a systematic review and meta-analysis, and was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guideline.11
Eligibility Criteria
Studies were included if they met the following criteria: adult patients aged 18 years or older who underwent primary or revision total hip arthroplasty (THA) for any indication; THA procedures involving cemented or uncemented implants, various surgical approaches, or different types of anesthesia; and studies reporting postoperative nerve injuries such as sciatic nerve palsy, femoral or obturator nerve injury, lateral femoral cutaneous nerve injury, superior gluteal nerve injury, or other peripheral neuropathies associated with THA. Eligible study designs included randomized controlled trials, cohort studies, case-control studies, and large case series involving at least five patients. Only articles published in English were considered.
Exclusion criteria included case reports or case series with fewer than five patients, cadaveric, animal, or biomechanical studies, conference abstracts without full-text availability, and studies that did not report nerve injury outcomes or were unrelated to THA.
Search Strategy
We performed the literature search using mainly two search engines: PubMed and Google Scholar. We have also included manual reference checks of relevant reviews and included studies to identify further eligible publications. The search strategy combined Medical Subject Headings (MeSH) and free-text terms related to THA and nerve injury.
Study Selection
All the records identified were imported into EndNote (Clarivate Analytics) to remove duplicates. Two independent reviewers searched titles and abstracts for potential eligible studies. Full text of the articles were subsequently acquired and examined for inclusion as per the predefined criteria. Disputes were settled by discussion or, where required, by convening the third reviewer.
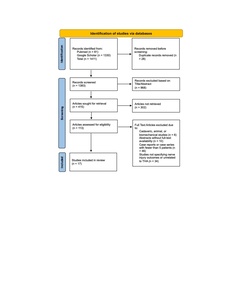
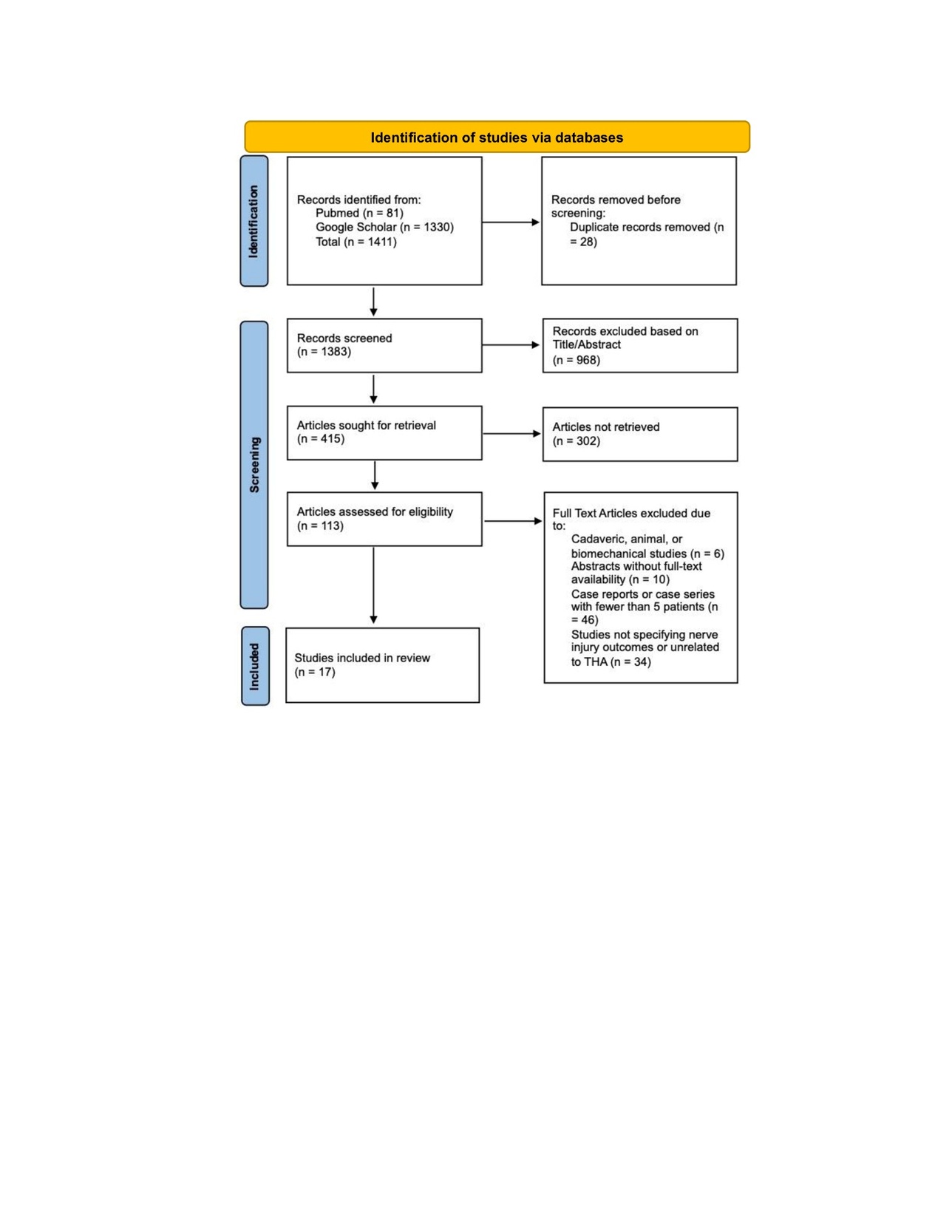
Study selection was described in the PRISMA flow diagram (Figure 1). Figure 1 represents the flow of records identified, screened, excluded (with reasons), and the total number of final studies included for this review.
Data Extraction
Extraction of data was conducted by two independent reviewers through the use of Microsoft Excel sheet. Any discrepancies between the reviewers were addressed through discussion until consensus was reached.
Quality Assessment of Included Studies
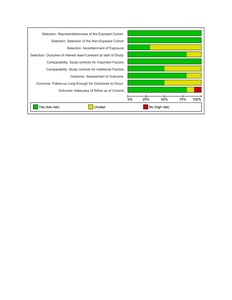
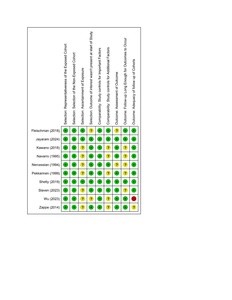
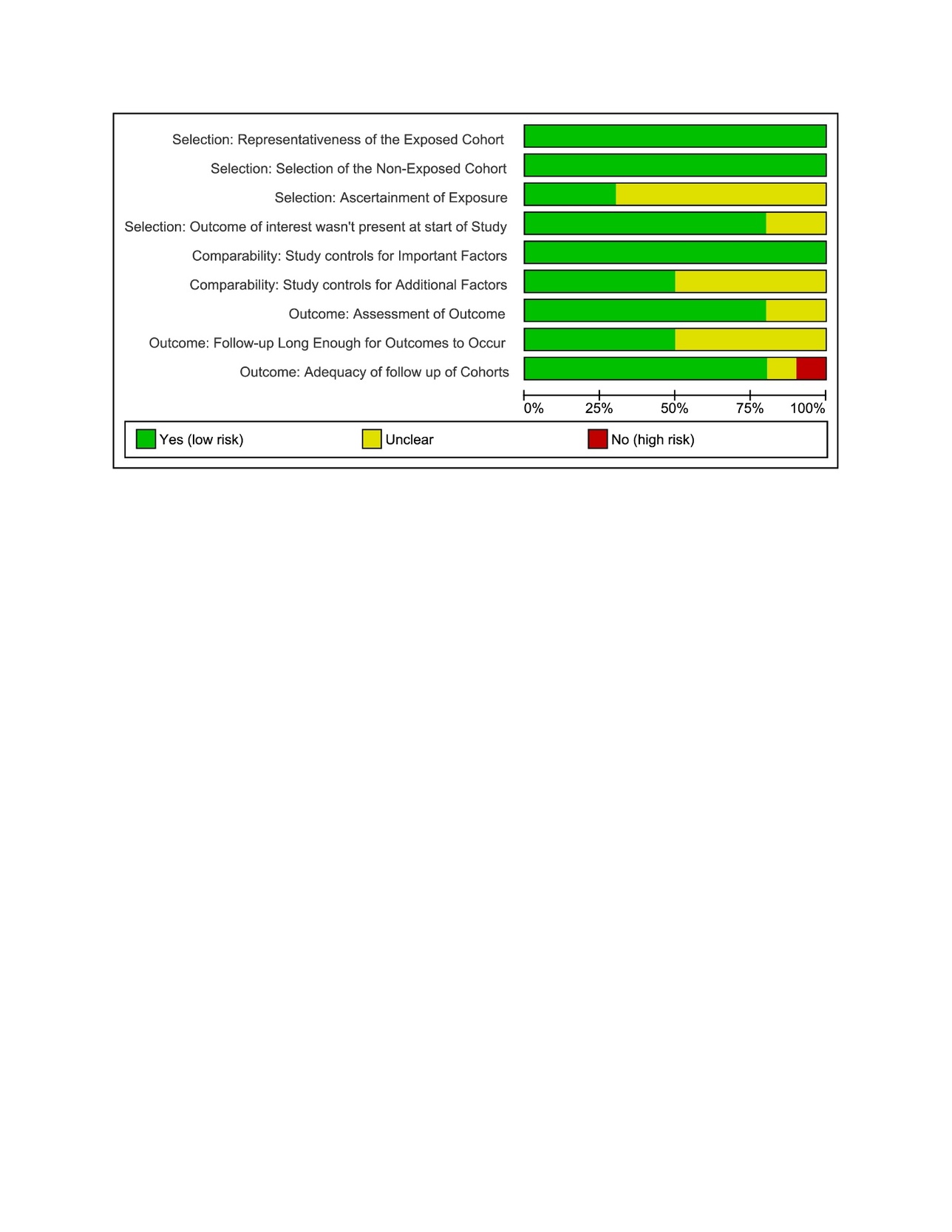
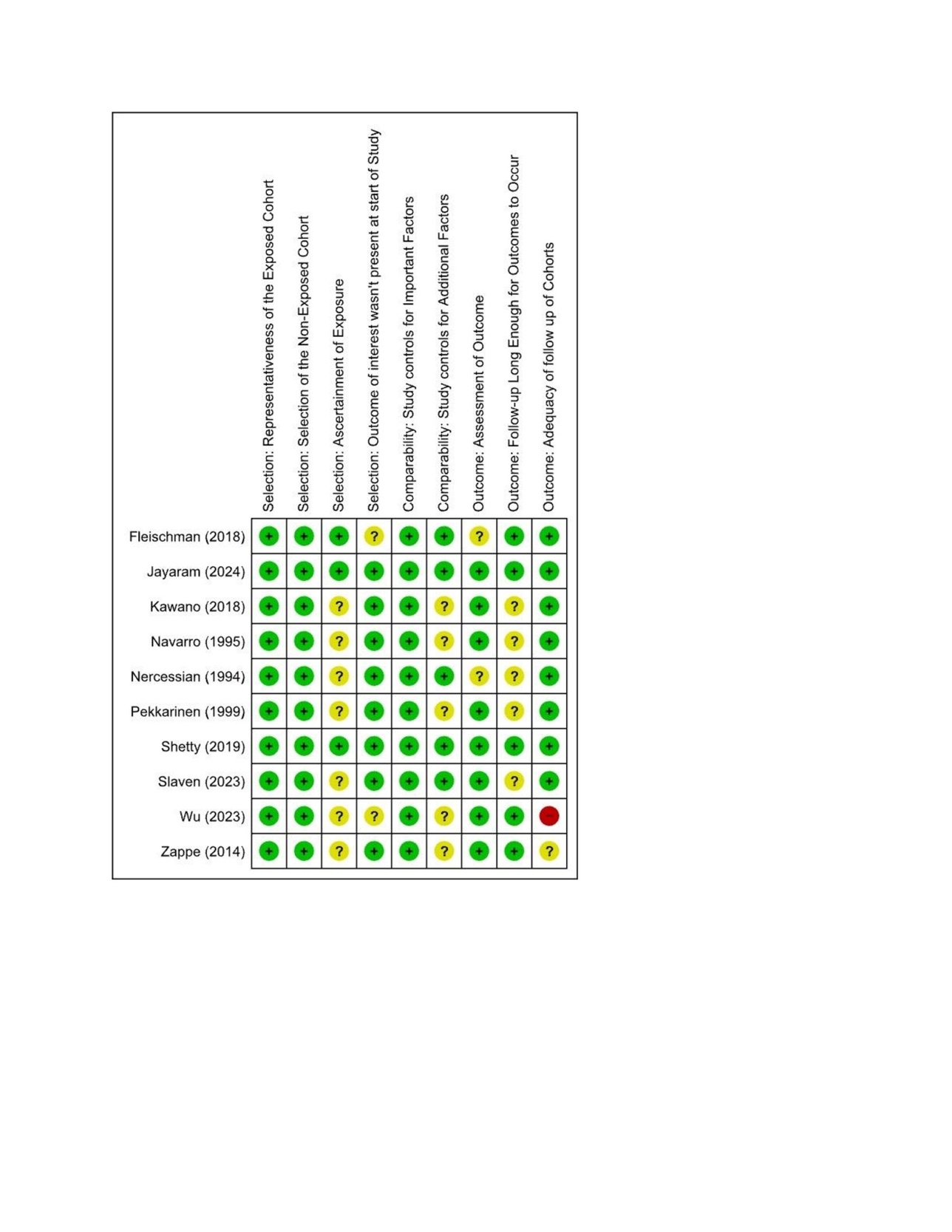
The quality of Cohort is assessed with NOS scale while for case series it was done with JBI checklist. The NOS cohort-quality table shows that the strongest evidence comes from large, well-designed retrospective cohorts such as Jayaram (2024) and Shetty (2019), both scoring 9/9 and rated as high quality. Studies like Slaven (2023), Fleischman (2018), Zappe (2014), Pekkarinen (1999), Navarro (1995), and Nercessian (1994) fall into the moderate category (scores 6–7), mainly limited by comparability issues and incomplete outcome reporting. Older studies were expectedly lower in methodological quality (Figure 2 & 3).
The JBI case series table further clarifies quality variation among non-comparative designs. The studies like Regev (2015) and Farrell (2005) demonstrate stronger methodological structure, scoring 8–9/10 due to clear inclusion criteria, consistent diagnostic assessment, and detailed clinical reporting. Other studies such as Patton (2018), Kyriacou (2013), and Butt (2005) were in the moderate range, which is often limited by incomplete follow-up, partial reporting of outcomes, or unclear participant inclusion processes. The earliest reports (Edwards 1987, Johanson 1983) scored lowest, which showed historical limitations in data collection and reporting standards (Table S1).
Results
Our review included 17 studies with a wide range of the research designs from the large retrospective database analyses to small case series. Most studies were conducted in the USA. The largest dataset came from Jayaram (2024), who analyzed more than 750,000 THA cases, while the smallest came from focused nerve-injury referral cohorts such as Wu (2023) and Regev (2015), each reporting 21 and 12 cases, respectively. The study periods varied greatly, with some covered the short windows of 1–5 years. Some of the other studies spanned over decades which included Johanson (1983) and Farrell (2005) as they included surgeries from the early 1970s onward. Follow-up durations also differed, which ranged from no formal follow-up reported (Kawano 2018, Kyriacou 2013) to long-term monitoring of 5–8 years in older orthopedic series. There are many studies which originated from tertiary referral centers, particularly those reporting only nerve injury cases (Wu 2023, Kyriacou 2013). The sample sizes across the retrospective cohorts varied widely from a few thousand (Zappe 2014; Kawano 2018) to tens of thousands (Shetty 2019; Farrell 2005). Overall, the number of the nerve injury cases ranged from 6 to over 2,600, which showed a mix of large epidemiologic reports and highly focused clinical series (Table 1).
Table 2 shows that the age of the patient at the time of nerve injury after THA generally fell between the late 50s and mid-60s, with most cohorts reporting mean ages of 58–65 years, such as Jayaram (2024),12 Slaven (2023),13 and Wu (2023).14 The distribution of gender showed that there is a consistent predominance of female patients, particularly in Kawano (2018),16 where all 14 affected patients were women. BMI values were commonly in the overweight range, with mean values around 28–30 kg/m² in several cohorts, including Slaven (2023)13 and Fleischman (2018).18 The degenerative conditions such as osteoarthritis were the most frequent comorbidities and underlying diagnoses, which is reported across multiple studies including Shetty (2019),15 Farrell (2005),22 and Navarro (1995).25 The developmental dysplasia of the hip (DDH) appeared often as a preoperative deformity, particularly noted in Kawano (2018),16 Farrell (2005),22 and Nercessian (1994).26 Most nerve-injury cases occurred after the primary THA, although several studies included revision procedures, such as Zappe (2014)20 and Pekkarinen (1999).24 The laterality was rarely highlighted, but when reported, injuries affected both sides as reported in Slaven (2023)13 and Navarro (1995).25
Table 3 shows the surgical parameters which reported that posterior or posterolateral approach was most frequently reported, used exclusively in several studies such as Shetty (2019),15 Kawano (2018),16 and Butt (2005).23 Direct anterior approaches were also common in recent work, especially in Slaven (2023)13 and Patton (2018).17 The lengthening of the limb emerged repeatedly as a relevant factor, with increases ranging from 6–20 mm in Shetty (2019)15 to more than 2 cm in several older series such as Farrell (2005)22 and Pekkarinen (1999).24 Intraoperative nerve monitoring was rarely used, except in Regev (2015)19 and selectively in Pekkarinen (1999).24 Common intraoperative complications included hematoma, retractor injury, cement extrusion, and direct nerve trauma, especially in older surgical cohorts like Edwards (1987)27 and Johanson (1983).28
Iatrogenic nerve injuries after THA most commonly affected the sciatic, peroneal, and femoral nerves, and the peroneal division repeatedly identified as the most vulnerable branch which is reported by Kawano (2018),16 Farrell (2005),22 and Pekkarinen (1999).24 Most of the injuries presented immediately postoperatively, especially when related to the traction, limb lengthening, or retractor pressure which is reported in Zappe (2014)20 and Edwards (1987).27 The delayed-onset cases were typically linked to structural causes such as prominent screws, hematoma, or the pseudotumor as documented in Wu (2023).14 The clinical manifestations of these injuries included foot drop, quadriceps weakness, sensory deficits, and neuropathic pain. Notably, the severity of these injuries commonly assessed with MRC grading, Sunderland classification, or EMG/NCS. The diagnostic confirmation frequently relied on MRI and electrodiagnostic studies, especially for the complex or progressive neuropathies. The intraoperative contributors of these injuries such as hematoma (Butt 200523), excessive limb lengthening (Johanson 198328), and direct mechanical trauma (Navarro 199525) were repeatedly observed (Table 4). In term of the management strategies, they were managed mainly by conservative approach, with focused on observation, physiotherapy, and orthotic support as seen in Slaven (2023)13 and Patton (2018),17 while surgery (e.g., hematoma evacuation, neurolysis, or component revision) was reserved for structural compression, progressive deficits, or absent recovery, as reported by Wu (2023)14 and Farrell (2005)22 (Table S2).
Table 5 shows that recovery after nerve injury following THA is highly variable and it ranged from excellent outcomes with the full return of function to the substantial long-term deficits. Notably, higher recovery rates were reported in cohorts with the isolated femoral or mild peroneal palsies, such as Slaven (2023),13 where 58–60% achieved full recovery within 5–8 months, and in Kawano (2018),16 where 71% recovered within 1–36 months. In contrast, the studies dominated by severe sciatic traction injuries which included Farrell (2005)22 and Johanson (1983)28 showed much poorer outcomes, with 33–79% of patients experienced persistent long-term deficits. The sensory symptoms were more resistant to recovery than the motor deficits; Fleischman (2018)18 reported sensory persistence in over 80% of cases despite 75% motor improvement. The early intervention clearly improved prognosis as a study by Butt (2005)23 showed complete recovery in all patients undergoing early hematoma evacuation, while delayed or the conservative approaches often resulted in the chronic disability. Orthotic use was common in peroneal palsies (e.g., Slaven 202313; Farrell 200522). Across studies, no mortality was attributed to nerve injury, but long-term functional impairment remained a significant concern, particularly in sciatic nerve injuries.
Meta-Analysis to Synthesize Quantitative Evidence
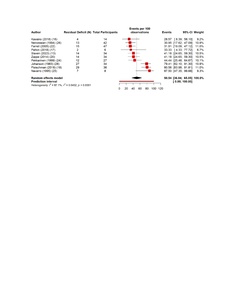
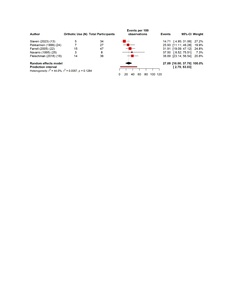
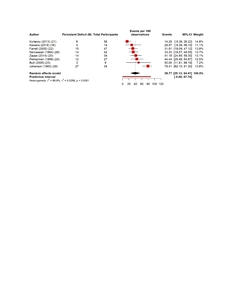
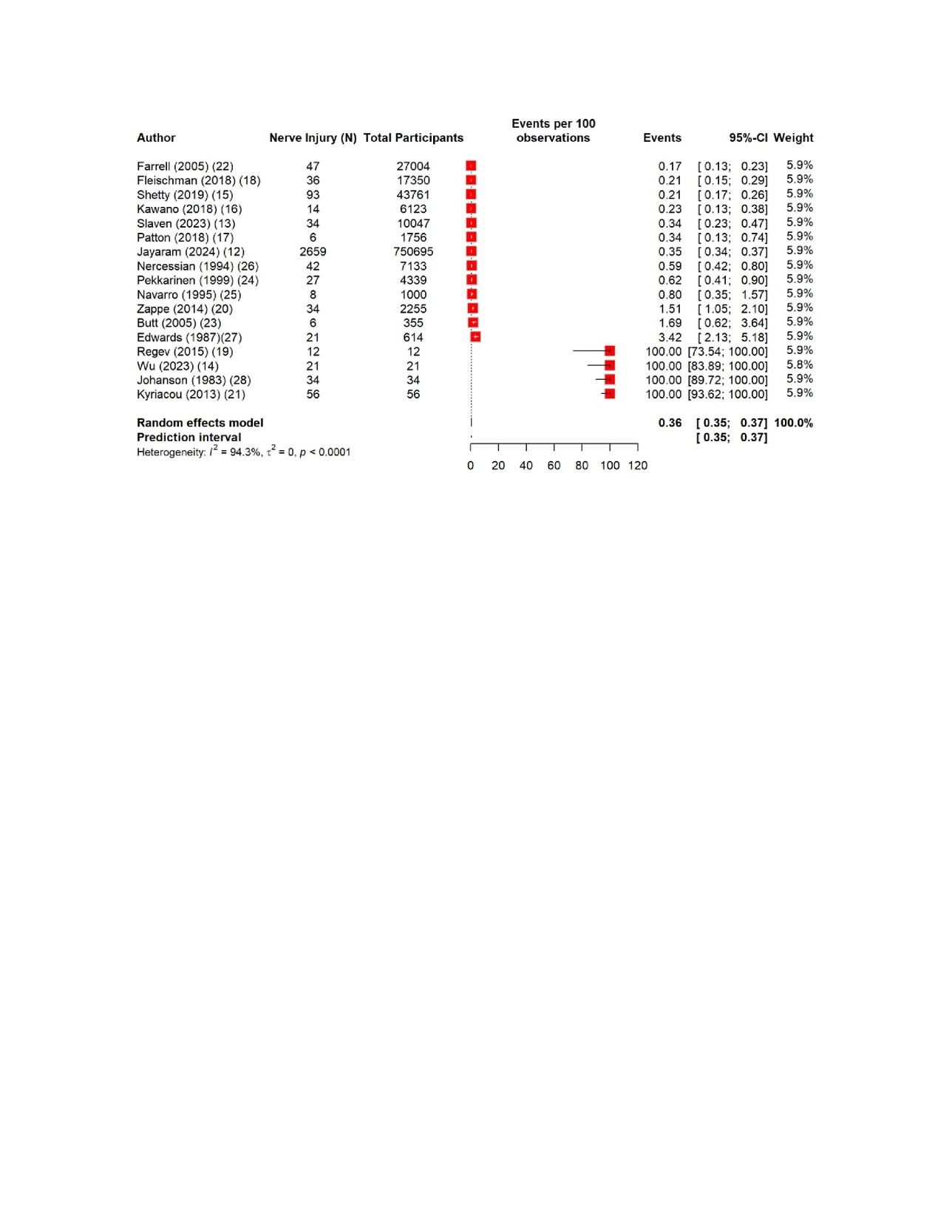
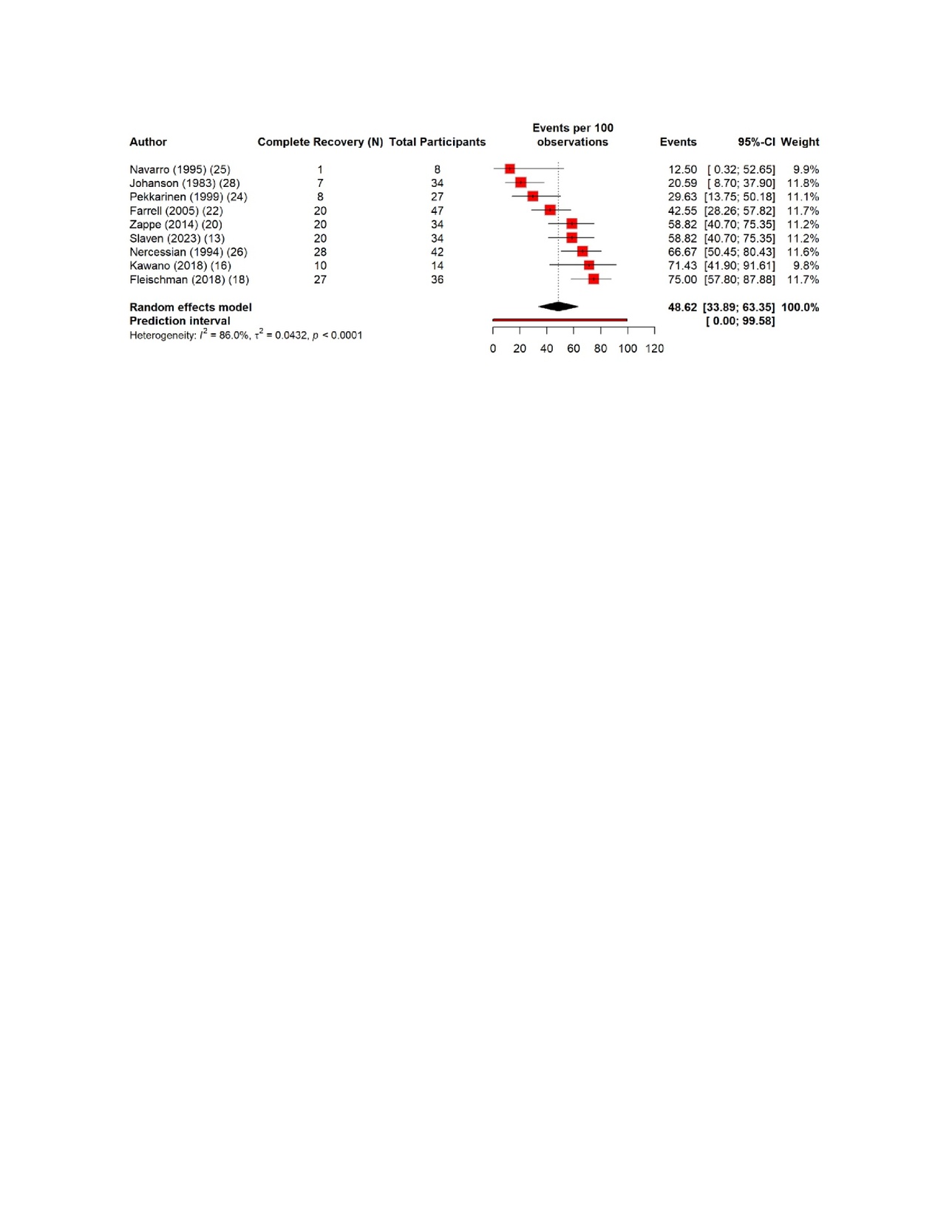
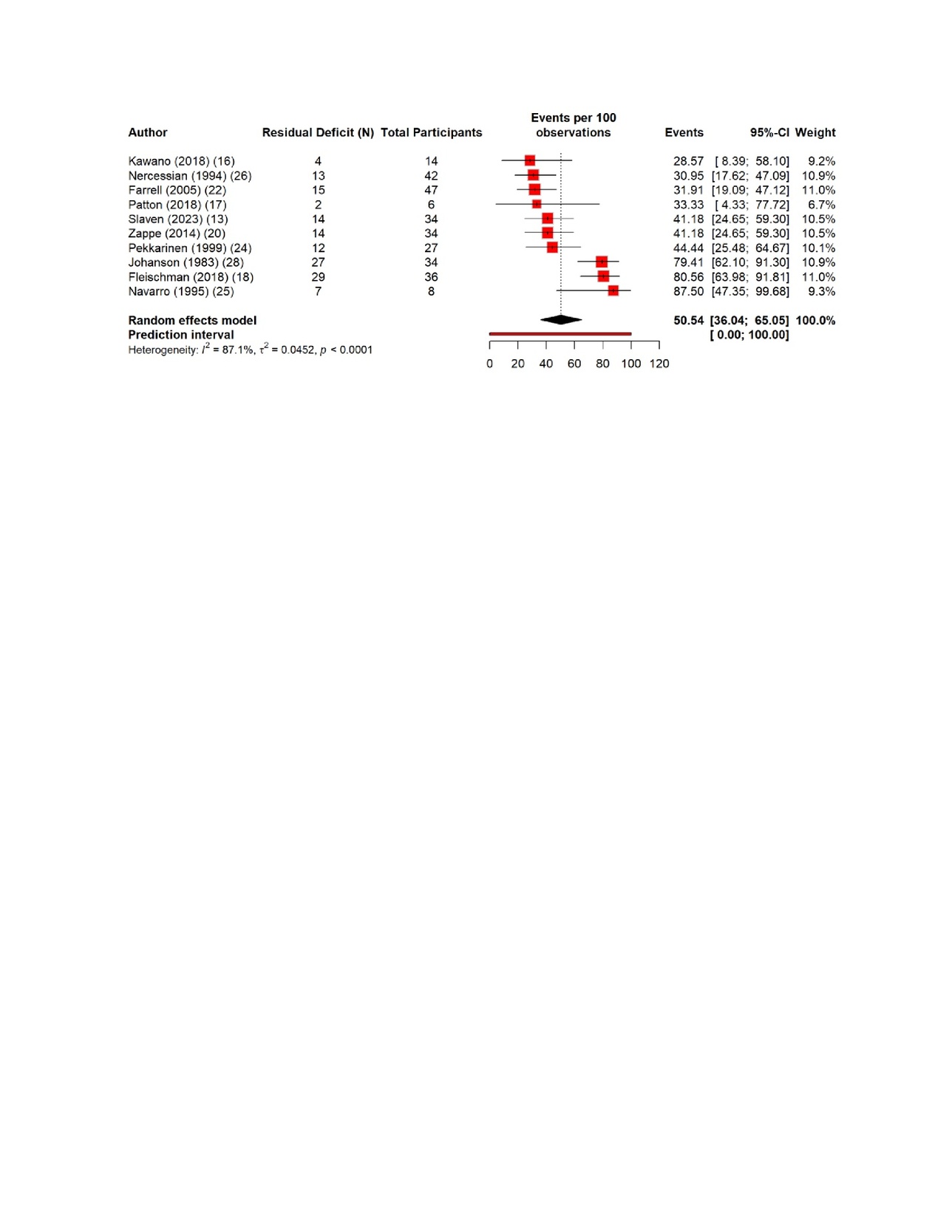
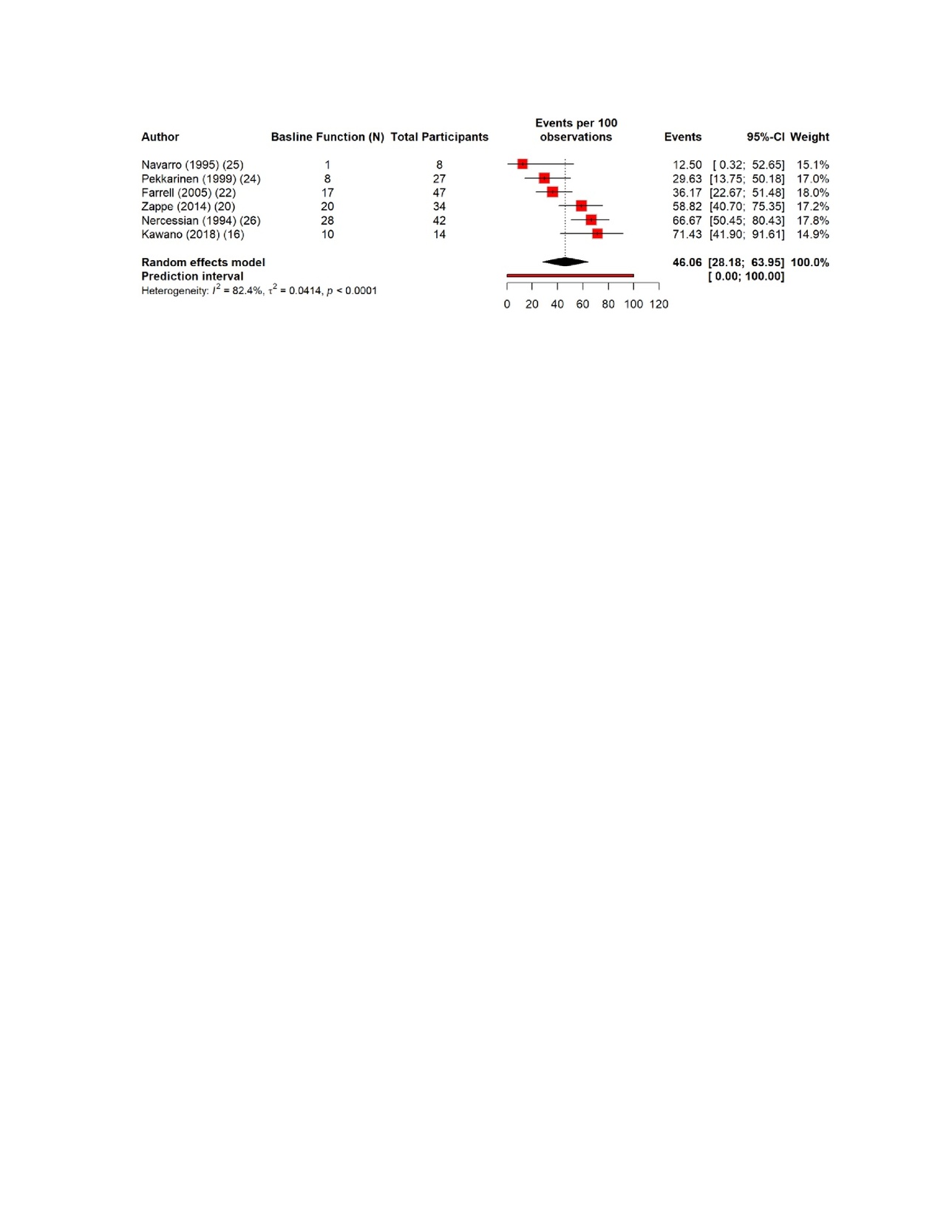
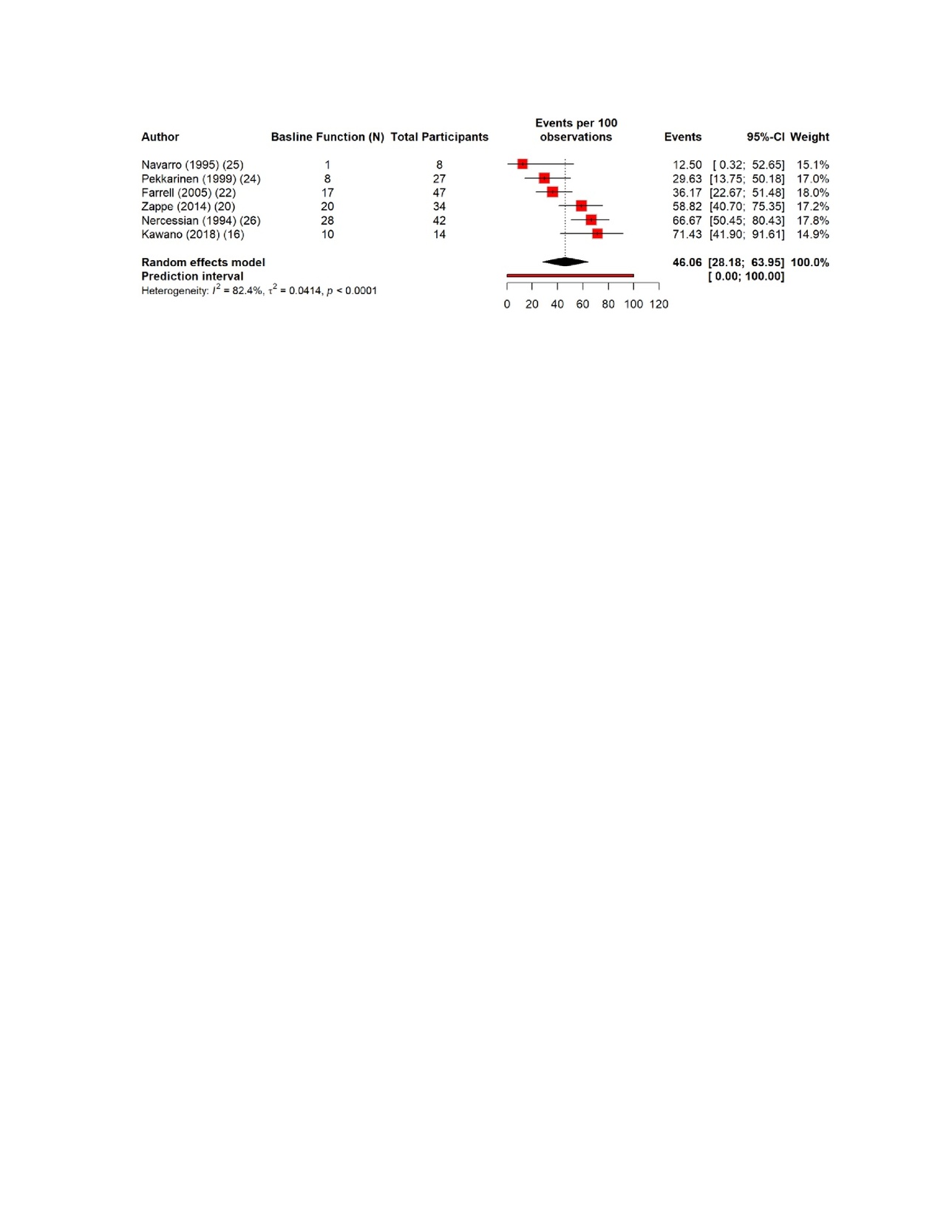
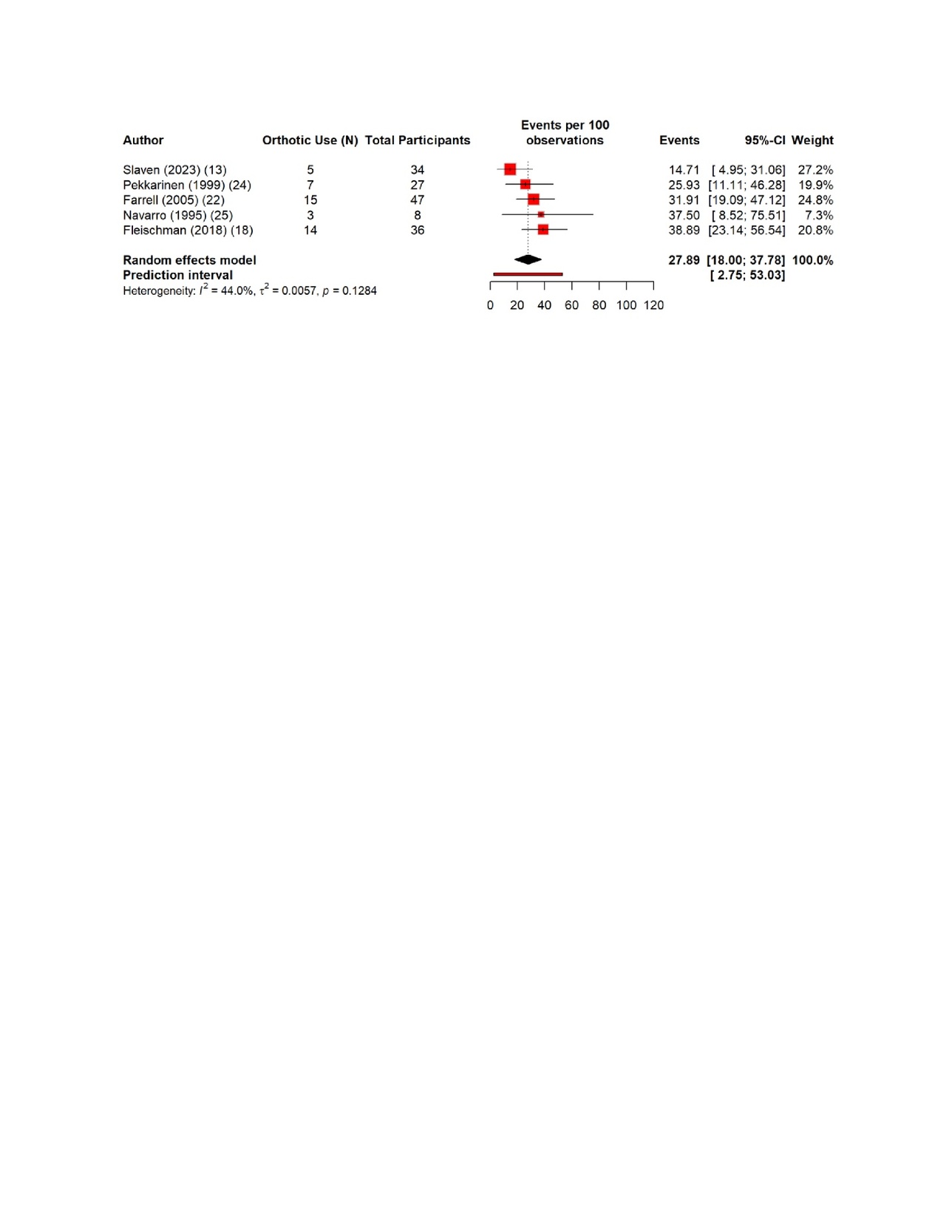
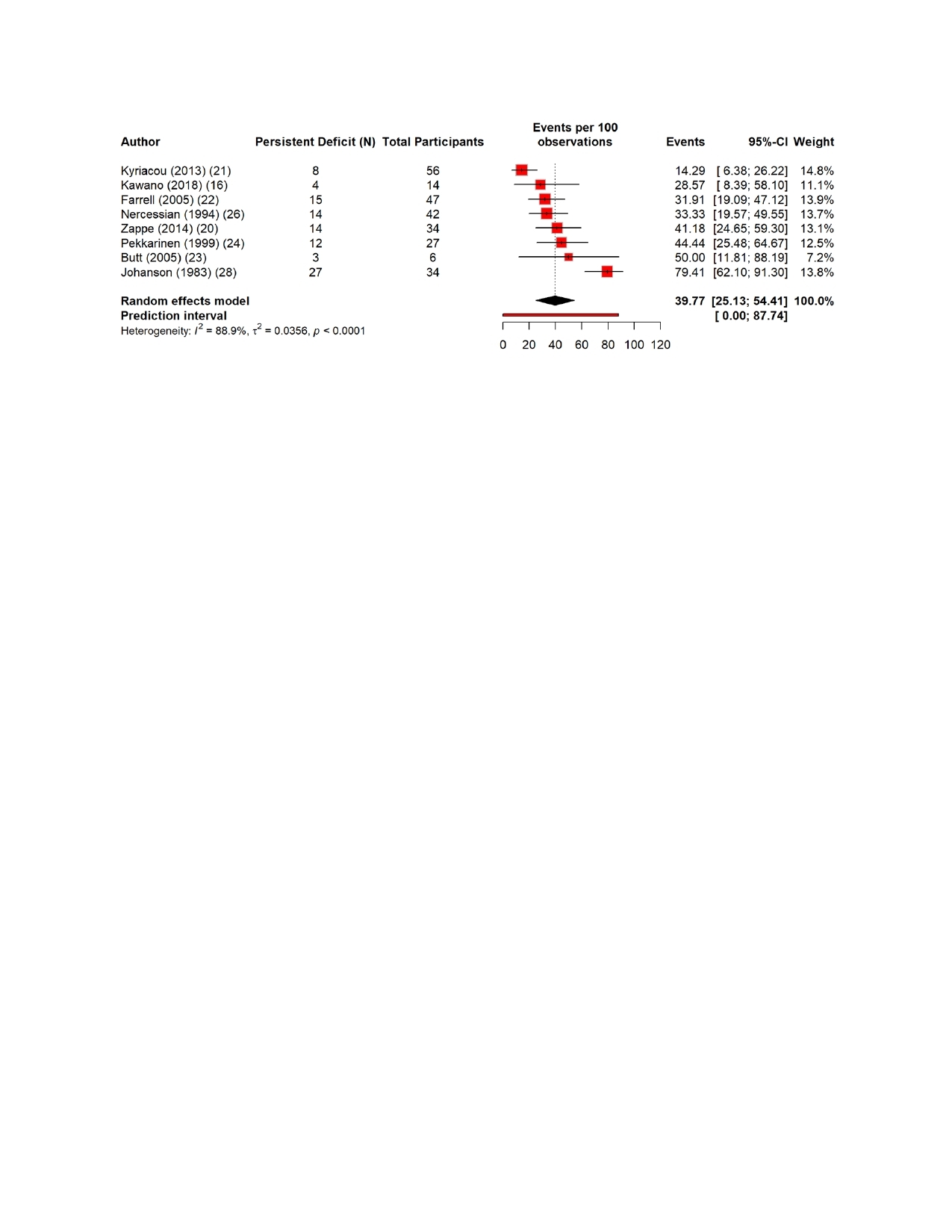
The pooled estimates show clear patterns in the burden and outcomes of the nerve injury after THA. The overall incidence of nerve injury was 0.36% (95% CI 0.35–0.637%), with extremely high heterogeneity (Figure 4). The complete recovery occurred in 48.6% of patients (95% CI 33.9–63.3%), while the residual deficits were slightly more common at 50.5% (95% CI 36.0–65.0%) (Figures 5 and 6). The reoperation for nerve injury showed that there is a pooled rate of 33.1% (95% CI 4.2–62.0%), reflecting wide variation between studies (Figure 7). The return to baseline function of injured nerve was achieved in 46.1% of cases (95% CI 28.2–63.9%), which indicated that many patients improve but do not fully regain pre-injury function (Figure 8). Orthotic use was required in 27.9% of patients (95% CI 18.0–37.8%), most commonly due to persistent foot drop or dorsiflexion weakness (Figure 9). Lastly, the permanent neurologic deficits were present in 39.8% of injuries (95% CI 25.1–54.4%), which highlighted the significant long-term impact of these complications (Figure 10).
Furthermore, Table S3 shows the pooled predictors of the nerve injury after THA. Only the presence of the comorbidities emerged as a statistically significant predictor, with higher comorbidity burden associated with the increased risk of nerve injury after THA (estimate 0.75, 95% CI −1.38 to −0.12, p = 0.019) which indicted the meaningful contribution to postoperative nerve vulnerability. The preoperative deformity showed a borderline association (estimate −0.57, p = 0.077), which suggested that anatomical abnormalities may increase risk, though evidence remains uncertain. The lateral positioning also trended toward increased risk (estimate 0.54, p = 0.097), which showed the possible mechanical or traction-related effect. Other variables which included surgical approach (anterior, posterior, mixed), intraoperative monitoring, supine positioning, surgeon experience, and intraoperative complications did not reach significance.
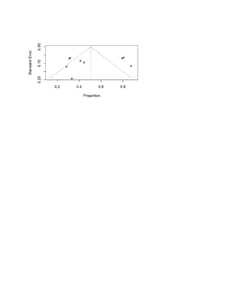
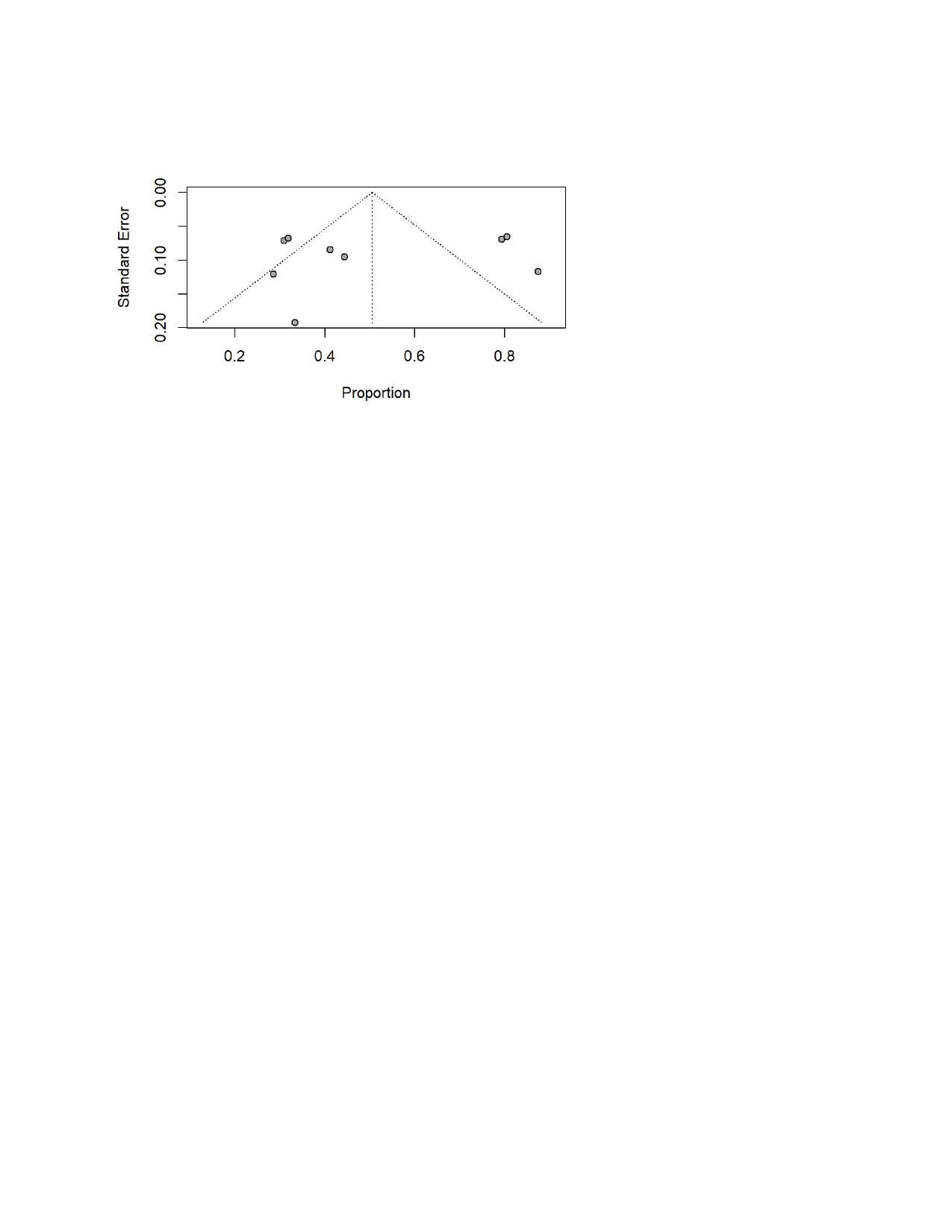
Figure 10 shows that funnel plot used to assess the publication bias in studies reporting nerve injury following THA. The plot demonstrates noticeable asymmetry, with the several studies clustering on the left side of the mean proportion and fewer appeared on the right, especially among those with the smaller standard errors. This pattern suggests a potential bias toward the publishment of the studies that report higher rates of permanent deficits. The spread of points outside the triangular pseudo-confidence region also reflects substantial heterogeneity across included studies, which is consistent with the high I² values observed in the meta-analysis. Although funnel plot asymmetry can arise from true clinical variability, the degree of imbalance seen here indicates that publication bias which likely favored the studies with more pronounced or clinically concerning outcomes cannot be ruled out.
Discussion
Total hip arthroplasty (THA) is a widely performed and highly effective procedure that restores mobility and reduces the pain for patients with degenerative joint disease.29 Although it is considered as a safe procedure, nerve injury remains a meaningful postoperative concern that can delay recovery and impair the long-term function.30 These injuries are more likely in individuals with the complex hip anatomy or the history of previous surgery and most often involve the sciatic or the femoral nerves. The mechanisms of these injuries included traction, compression, or direct surgical trauma. Because the reported risks vary widely across studies, there is a need for high-quality synthesized evidence in order to clarify true incidence and guide prevention. This review aims to examine the burden, risk factors, clinical characteristics, management, and outcomes of nerve injuries after total hip arthroplasty (THA).
Across 17 studies, our findings highlight that nerve injury is an uncommon but clinically meaningful complication, with wide variation in reported incidence, mechanisms, and long-term outcomes. The pooled incidence was 0.36% (95% CI 0.35–0.37%), which aligned with previous literature which reported that the rates of nerve injury after THA ranged between 0.1% and 3%, depending on sample size, diagnostic rigor, and surgical approach (Brown et al. 2008).31 Another study by Knebel et al. (2025) which consistently demonstrated low incidence rate (0.7%) but important risk levels, particularly in patients with complex anatomy or high comorbidity burden.32
There is a key pattern which revealed in the included studies is the predominance of injuries affecting the sciatic and peroneal nerves, which is followed by femoral neuropathies. These findings aligned with earlier work by Park et al. (2013) who identified the peroneal division of the sciatic nerve as particularly vulnerable because of its fixed anatomic position and susceptibility to traction during hip dislocation or limb lengthening.33 Our review confirms this pattern, with the peroneal division being the most frequently reported site of injury in several series (Kawano 2018, Farrell 2005, Pekkarinen 1999).16,22,24 These injuries often manifested immediately postoperatively, which is also reported in older orthopedic literature which showed that traction- and retractor-related palsies typically present upon awakening from anesthesia.34 Moreover, the delayed-onset neuropathies is largely caused by structural issues such as prominent screws, hematomas, or pseudotumors and they are also well-documented in prior study by Balaji et al. (2019) which emphasized the importance of postoperative imaging when deficits evolve over days to weeks.35
There are several surgical factors who played a significant role across the evaluated studies. Posterior or the posterolateral approaches were the most commonly reported, which reflects historical practice patterns. Although some earlier literature suggested higher sciatic nerve risk with posterior approaches,36 but more recent work has shown mixed results, which indicated that surgeon technique and patient anatomy may be more influential than approach alone.37 Limb lengthening also emerged as a recurring contributor to injury. Our findings show lengthening values ranging from 6 to more than 20 mm in several cohorts, which aligned with earlier studies that demonstrated a linear relationship between traction forces and nerve conduction impairment. Higuchi et al. (2015) documented greater-than-2-cm lengthening as a major predictor of persistent deficits, and this association persists in more modern series.38
Furthermore, the analysis of predictors revealed that comorbidity burden was the only statistically significant factor associated with increased risk of nerve injury. This finding is supported by existing evidence linking systemic illness such as diabetes, vascular disease, and inflammatory disorders to nerve vulnerability. Previous research by Sonawane et al. (2023) has indicated that preexisting neuropathic changes, microvascular compromise, and metabolic stress can reduce the threshold for intraoperative nerve damage.39 Although preoperative deformity also trended toward significance, its effect did not reach statistical threshold. However, the previous orthopedic literature consistently shows that dysplasia, posttraumatic deformities, and ankylosis increase the technical complexity of THA and therefore raise the risk of nerve injury.40 Our borderline results likely reflect heterogeneity in how deformities were defined and reported.
The management patterns were also consistent across studies, with most nerve injuries initially treated conservatively through observation, physiotherapy, and orthotic support. This aligned with established clinical practice guidelines recommending non-operative management for neuropraxias or mild axonotmesis. The surgery was reserved for structural or progressive deficits, such as hematoma evacuation, neurolysis, or revision surgery to address excessive limb lengthening or hardware-related compression. The strong outcomes seen in early hematoma evacuation cases support earlier recommendations from the peripheral nerve literature emphasizing rapid decompression for expanding hematomas.41 Neurolysis in chronic compression cases showed variable benefit, a finding which is consistent with previous microsurgical study by Grisdela et al. (2023) which reported partial improvement in most patients but limited sensory recovery due to long-standing axonal damage.42
Recovery outcomes varied widely among the included studies. Our pooled data showed that complete recovery occurred in 48.6% of patients, while residual deficits were present in 50.5%, and nearly 40% had permanent deficits. These findings aligned with historical orthopedic reports which noted that between one-third and one-half of patients with sciatic nerve palsies after THA have some degree of long-term functional loss. Similarly, a study by Zappe et al. (2014) shows that 50 % of the patients showed a complete recovery after 2 years.43 The motor recovery was generally more favorable than sensory recovery, which aligns with neurophysiological principles and prior studies showing that large motor fibers have better regenerative potential compared to smaller sensory fibers (Ali et al. 2019).44 The high rates of persistent sensory symptoms observed in Fleischman (2018), where over 80% retained some deficit, further reinforce this concept.18
Return to baseline function was achieved in 46% of cases, and orthotic use was required in nearly 28%, particularly for peroneal palsies causing persistent foot drop. These findings are consistent with longstanding observations that functional recovery lags behind pure muscle strength recovery, especially when gait abnormalities or proprioceptive loss persist (Buckthorpe et al. 2023).45 Encouragingly, none of the study reported mortality attributable to nerve injury, though the long-term functional burden remained significant.
Implications
There are several important clinical implications of our findings. Surgeons should recognize that nerve injury remains a substantial source of the functional decline after THA, especially in patients with the prior history of deformity, multiple comorbidities, or the complex anatomy. The careful preoperative planning, meticulous handling of the soft-tissue, and heightened awareness of limb lengthening are essential in order to reduce risk. The early identification of the neurological deficits can guide timely intervention, particularly in cases which required the hematoma evacuation or neurolysis. There is a need of clear patient counseling about expected recovery timelines and potential for the persistent deficits which will improve shared decision-making and postoperative expectations.
Conclusion
Our study shows that nerve injury after THA is very rare, but it remains a clinically important complication with the substantial functional consequences. Most of these injuries involved the sciatic, peroneal, and femoral nerves, with recovery patterns ranged from full resolution to persistent long-term deficits. The predictors of these injuries during THA included higher comorbidity burden and complex anatomy, while early recognition and timely intervention improve outcomes after injuries. These findings highlight the need for vigilant surgical technique, careful patient selection, and clear counseling to reduce risk and optimize recovery in THA patients.
Abbreviations
-
THA – Total Hip Arthroplasty
-
PNP – Peroneal Nerve Palsy
-
FNP – Femoral Nerve Palsy
-
DDH – Developmental Dysplasia of the Hip
-
EMG – Electromyography
-
AFO – Ankle–Foot Orthosis
Author Contributions
A.T., T.M., A.H., F.H., M.A., A.A., A.Q., and Z.J. contributed equally to this work. All authors made substantial contributions to the conception and design of the study, acquisition, analysis, and interpretation of data, drafting of the manuscript and critical revision for important intellectual content. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work, ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.